QUESTION IMAGE
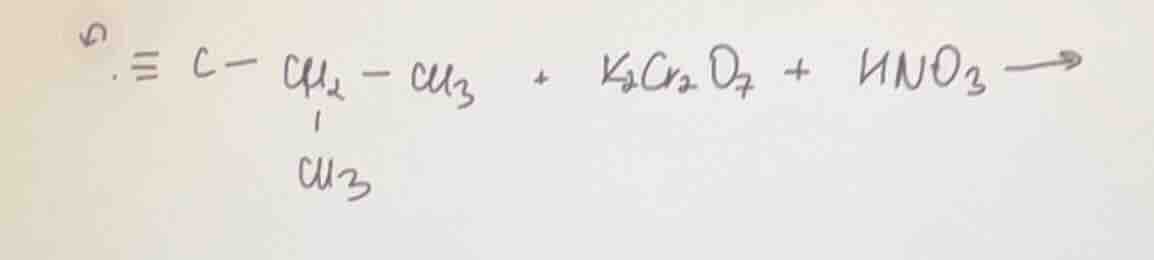
Question
≡c - ch₂ - ch₃ (with a ch₃ branch on the ch₂) + k₂cr₂o₇ + hno₃ →
To balance the redox reaction involving the organic compound (let's assume the organic compound is a hydrocarbon derivative, here we can analyze the oxidation states first), $\ce{K2Cr2O7}$ (where $\ce{Cr}$ is in +6 oxidation state) and $\ce{HNO3}$ (here $\ce{N}$ is in +5 oxidation state, and it may act as an acid or an oxidizing agent, but in this case, likely the $\ce{K2Cr2O7}$ is the main oxidizing agent for the organic compound).
Step 1: Identify Oxidation States of Key Atoms
- For the organic compound (let's denote the organic compound as $\ce{R}$ where $\ce{R}$ has a triple bond and alkyl groups: $\ce{≡C - CH2 - CH3}$ with a $\ce{CH3}$ branch, let's simplify the carbon chain. Let's consider the carbon atoms:
- The terminal alkyne carbon ($\ce{≡C -}$): Let's assume the oxidation state of $\ce{C}$ here. In alkanes, $\ce{C}$ has an oxidation state of -3 (in $\ce{CH4}$), in alkenes (double bond) maybe -2, in alkynes (triple bond) maybe -1? Wait, better to use the method of calculating oxidation state by considering the bonds. For a carbon atom bonded to other carbons and hydrogens: each $\ce{C - H}$ bond contributes +1 to $\ce{H}$ and -1 to $\ce{C}$; each $\ce{C - C}$ bond contributes 0 (since same electronegativity).
- Let's take the main chain: $\ce{≡C - CH2 - CH3}$ (with a $\ce{CH3}$ on the $\ce{CH2}$? Wait, the structure is $\ce{≡C - CH(CH3) - CH3}$? Maybe a typo, but let's proceed. Let's assume the organic compound is $\ce{HC≡C - CH(CH3) - CH3}$ (2 - methyl - 1 - butyne). Let's calculate oxidation state of $\ce{C}$:
- Terminal alkyne $\ce{C}$ (bonded to $\ce{H}$ and $\ce{C}$): $\ce{H - C≡C -}$: $\ce{H}$ is +1, so this $\ce{C}$ has oxidation state: let $x$ be oxidation state of $\ce{C}$, then $x + 1 + (x + 3) = 0$? Wait, no, for a $\ce{C}$ atom: sum of oxidation states of bonded atoms = 0 (for neutral compound). For $\ce{H - C≡C -}$: $\ce{H}$ is +1, the other $\ce{C}$ is bonded to this $\ce{C}$ (triple bond). So for the terminal $\ce{C}$ (bonded to $\ce{H}$ and $\ce{C}$): $x$ (oxidation state of $\ce{C}$) + 1 (from $\ce{H}$) + 3*(0) (from $\ce{C - C}$ triple bond? Wait, no, oxidation state is calculated as: for each bond to a more electronegative atom, $\ce{C}$ loses electrons; to less electronegative, gains. $\ce{H}$ is less electronegative than $\ce{C}$, so $\ce{C - H}$: $\ce{C}$ has -1, $\ce{H}$ has +1. $\ce{C - C}$: 0. So terminal alkyne $\ce{C}$ (bonded to $\ce{H}$ and $\ce{C}$): oxidation state = -1 (from $\ce{C - H}$) + 0 (from $\ce{C - C}$) = -1.
- The next $\ce{C}$ (in $\ce{-C≡C - CH(CH3) -}$): bonded to two $\ce{C}$ atoms (triple and single) and no $\ce{H}$? Wait, maybe the structure is $\ce{HC≡C - CH2 - CH3}$ with a $\ce{CH3}$ on the $\ce{CH2}$, so $\ce{HC≡C - CH(CH3) - CH3}$. Let's take the $\ce{C}$ in $\ce{-CH(CH3) -}$: bonded to two $\ce{C}$ atoms, one $\ce{H}$, and one $\ce{C}$ (from $\ce{CH3}$). So oxidation state: $x + 0$ (from $\ce{C - C}$) + 1 (from $\ce{C - H}$) + 0 (from $\ce{C - C}$) = 0? No, better to use the fact that in redox reactions with $\ce{K2Cr2O7}$, the organic compound (hydrocarbon or derivative) is oxidized to $\ce{CO2}$ (if fully oxidized) or carboxylic acids, etc. $\ce{K2Cr2O7}$ is reduced to $\ce{Cr^3+}$ (oxidation state +3).
Step 2: Write Half - Reactions
- Reduction Half - Reaction ($\ce{K2Cr2O7 -> Cr^3+}$):
$\ce{Cr2O7^{2 - } + 14H+ + 6e- -> 2Cr^3+ + 7H2O}$
(Each $\ce{Cr}$ goes from +6 to +3, so 3 electrons gained per $\ce{Cr}$; 2 $\ce{Cr}$ atoms, so 6 electrons gained total. Balanced with $\ce{H+}$ and $\ce{H2O}$ in acidic medium, as $\ce{HNO3}$ provides $\ce{…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To balance the redox reaction involving the organic compound (let's assume the organic compound is a hydrocarbon derivative, here we can analyze the oxidation states first), $\ce{K2Cr2O7}$ (where $\ce{Cr}$ is in +6 oxidation state) and $\ce{HNO3}$ (here $\ce{N}$ is in +5 oxidation state, and it may act as an acid or an oxidizing agent, but in this case, likely the $\ce{K2Cr2O7}$ is the main oxidizing agent for the organic compound).
Step 1: Identify Oxidation States of Key Atoms
- For the organic compound (let's denote the organic compound as $\ce{R}$ where $\ce{R}$ has a triple bond and alkyl groups: $\ce{≡C - CH2 - CH3}$ with a $\ce{CH3}$ branch, let's simplify the carbon chain. Let's consider the carbon atoms:
- The terminal alkyne carbon ($\ce{≡C -}$): Let's assume the oxidation state of $\ce{C}$ here. In alkanes, $\ce{C}$ has an oxidation state of -3 (in $\ce{CH4}$), in alkenes (double bond) maybe -2, in alkynes (triple bond) maybe -1? Wait, better to use the method of calculating oxidation state by considering the bonds. For a carbon atom bonded to other carbons and hydrogens: each $\ce{C - H}$ bond contributes +1 to $\ce{H}$ and -1 to $\ce{C}$; each $\ce{C - C}$ bond contributes 0 (since same electronegativity).
- Let's take the main chain: $\ce{≡C - CH2 - CH3}$ (with a $\ce{CH3}$ on the $\ce{CH2}$? Wait, the structure is $\ce{≡C - CH(CH3) - CH3}$? Maybe a typo, but let's proceed. Let's assume the organic compound is $\ce{HC≡C - CH(CH3) - CH3}$ (2 - methyl - 1 - butyne). Let's calculate oxidation state of $\ce{C}$:
- Terminal alkyne $\ce{C}$ (bonded to $\ce{H}$ and $\ce{C}$): $\ce{H - C≡C -}$: $\ce{H}$ is +1, so this $\ce{C}$ has oxidation state: let $x$ be oxidation state of $\ce{C}$, then $x + 1 + (x + 3) = 0$? Wait, no, for a $\ce{C}$ atom: sum of oxidation states of bonded atoms = 0 (for neutral compound). For $\ce{H - C≡C -}$: $\ce{H}$ is +1, the other $\ce{C}$ is bonded to this $\ce{C}$ (triple bond). So for the terminal $\ce{C}$ (bonded to $\ce{H}$ and $\ce{C}$): $x$ (oxidation state of $\ce{C}$) + 1 (from $\ce{H}$) + 3*(0) (from $\ce{C - C}$ triple bond? Wait, no, oxidation state is calculated as: for each bond to a more electronegative atom, $\ce{C}$ loses electrons; to less electronegative, gains. $\ce{H}$ is less electronegative than $\ce{C}$, so $\ce{C - H}$: $\ce{C}$ has -1, $\ce{H}$ has +1. $\ce{C - C}$: 0. So terminal alkyne $\ce{C}$ (bonded to $\ce{H}$ and $\ce{C}$): oxidation state = -1 (from $\ce{C - H}$) + 0 (from $\ce{C - C}$) = -1.
- The next $\ce{C}$ (in $\ce{-C≡C - CH(CH3) -}$): bonded to two $\ce{C}$ atoms (triple and single) and no $\ce{H}$? Wait, maybe the structure is $\ce{HC≡C - CH2 - CH3}$ with a $\ce{CH3}$ on the $\ce{CH2}$, so $\ce{HC≡C - CH(CH3) - CH3}$. Let's take the $\ce{C}$ in $\ce{-CH(CH3) -}$: bonded to two $\ce{C}$ atoms, one $\ce{H}$, and one $\ce{C}$ (from $\ce{CH3}$). So oxidation state: $x + 0$ (from $\ce{C - C}$) + 1 (from $\ce{C - H}$) + 0 (from $\ce{C - C}$) = 0? No, better to use the fact that in redox reactions with $\ce{K2Cr2O7}$, the organic compound (hydrocarbon or derivative) is oxidized to $\ce{CO2}$ (if fully oxidized) or carboxylic acids, etc. $\ce{K2Cr2O7}$ is reduced to $\ce{Cr^3+}$ (oxidation state +3).
Step 2: Write Half - Reactions
- Reduction Half - Reaction ($\ce{K2Cr2O7 -> Cr^3+}$):
$\ce{Cr2O7^{2 - } + 14H+ + 6e- -> 2Cr^3+ + 7H2O}$
(Each $\ce{Cr}$ goes from +6 to +3, so 3 electrons gained per $\ce{Cr}$; 2 $\ce{Cr}$ atoms, so 6 electrons gained total. Balanced with $\ce{H+}$ and $\ce{H2O}$ in acidic medium, as $\ce{HNO3}$ provides $\ce{H+}$.)
- Oxidation Half - Reaction (Organic Compound -> Oxidized Product, e.g., $\ce{CO2}$ or Carboxylic Acid):
Let's assume the organic compound is $\ce{HC≡C - CH(CH3) - CH3}$ (molar mass and carbon count: 5 $\ce{C}$ atoms). Let's say it's oxidized to $\ce{CO2}$ (full oxidation). Each $\ce{C}$ in the organic compound: let's find the average oxidation state. In the organic compound, let's calculate the oxidation state of $\ce{C}$:
For a $\ce{C}$ atom in $\ce{HC≡C -}$: as above, -1; in $\ce{-CH(CH3) -}$: let's calculate: bonded to two $\ce{C}$ (0 each), one $\ce{H}$ (+1), and one $\ce{C}$ (0), so $x + 0 + 1 + 0 = 0$? No, better to use the number of electrons lost. Each $\ce{C}$ in the organic compound (5 $\ce{C}$ atoms) is oxidized to $\ce{CO2}$ (where $\ce{C}$ is +4). So for each $\ce{C}$ atom: change in oxidation state = $4 - x$ (where $x$ is initial oxidation state). Let's take the initial oxidation state of $\ce{C}$ in the organic compound as an average. Let's assume the organic compound has 5 $\ce{C}$ atoms. Let the initial average oxidation state of $\ce{C}$ be $x$, final in $\ce{CO2}$ is +4. So total electrons lost per molecule of organic compound: $5*(4 - x)$. From the reduction half - reaction, each $\ce{Cr2O7^{2 - }}$ gains 6 electrons.
Let's assume the organic compound has $n$ $\ce{C}$ atoms (let's say $n = 5$). Let's denote the organic compound as $\ce{R}$ with $n$ $\ce{C}$ atoms. The oxidation half - reaction (simplified for 5 $\ce{C}$ atoms):
$\ce{HC≡C - CH(CH3) - CH3 + H2O -> 5CO2 + (1 + 2 + 1 + 3)H+ + ne-}$ (wait, better to count $\ce{H}$ atoms: the organic compound has $\ce{H}$ atoms: in $\ce{HC≡C -}$: 1 $\ce{H}$; in $\ce{-CH(CH3) -}$: 1 $\ce{H}$; in $\ce{-CH3}$: 3 $\ce{H}$; and the other $\ce{-CH3}$: 3 $\ce{H}$. Total $\ce{H}$: $1 + 1 + 3 + 3 = 8$? Wait, no, structure: $\ce{HC≡C - CH(CH3) - CH3}$: $\ce{H - C≡C - C(CH3)H - CH3}$: so $\ce{H}$ atoms: 1 (terminal alkyne) + 1 (in $\ce{-CH(CH3) -}$) + 3 (in $\ce{-CH3}$) + 3 (in the other $\ce{-CH3}$) = 8 $\ce{H}$ atoms. So the organic compound is $\ce{C5H8}$.
Oxidation of $\ce{C5H8}$ to $\ce{CO2}$:
$\ce{C5H8 + 10H2O -> 5CO2 + 28H+ + 28e-}$
(Balancing $\ce{C}$: 5 $\ce{C}$ in reactant, 5 $\ce{CO2}$. Balancing $\ce{H}$: 8 $\ce{H}$ in reactant, so 28 $\ce{H+}$ (since $5*2*H2O$? Wait, no: $\ce{C5H8 + H2O -> CO2 + H+}$. Let's do it properly:
$\ce{C5H8 -> 5CO2 + 8H+ + ne-}$
Balance $\ce{O}$: add $\ce{H2O}$ to left: $\ce{C5H8 + 10H2O -> 5CO2 + 28H+ + ne-}$
Balance $\ce{H}$: left has 8 + 20 = 28 $\ce{H}$; right has 28 $\ce{H+}$, so balanced.
Balance charge: left charge: 0; right charge: $28(+1) + ne-$. So $0 = 28 + (-n)$ → $n = 28$. So each $\ce{C5H8}$ loses 28 electrons.
Step 3: Balance Electrons in Half - Reactions
- Reduction: 6 electrons gained per $\ce{Cr2O7^{2 - }}$
- Oxidation: 28 electrons lost per $\ce{C5H8}$
To balance electrons, find the least common multiple of 6 and 28, which is 84.
- Multiply reduction half - reaction by 14: $\ce{14Cr2O7^{2 - } + 196H+ + 84e- -> 28Cr^3+ + 98H2O}$
- Multiply oxidation half - reaction by 3: $\ce{3C5H8 + 30H2O -> 15CO2 + 84H+ + 84e-}$
Step 4: Combine Half - Reactions
Add the two half - reactions:
$\ce{14Cr2O7^{2 - } + 196H+ + 84e- + 3C5H8 + 30H2O -> 28Cr^3+ + 98H2O + 15CO2 + 84H+ + 84e-}$
Simplify (cancel electrons, $\ce{H+}$, $\ce{H2O}$):
- Electrons: cancel.
- $\ce{H+}$: $196H+ - 84H+ = 112H+$
- $\ce{H2O}$: $98H2O - 30H2O = 68H2O$
So:
$\ce{3C5H8 + 14Cr2O7^{2 - } + 112H+ -> 15CO2 + 28Cr^3+ + 68H2O}$
Now, add $\ce{K+}$ (from $\ce{K2Cr2O7}$) and $\ce{NO3-}$ (from $\ce{HNO3}$) to balance the equation in terms of spectator ions (since $\ce{HNO3}$ is a strong acid, providing $\ce{H+}$ and $\ce{NO3-}$; $\ce{K2Cr2O7}$ provides $\ce{K+}$ and $\ce{Cr2O7^{2 - }}$).
From $\ce{14K2Cr2O7}$, we have $28K+$ and $14Cr2O7^{2 - }$.
From $\ce{HNO3}$, we need $112H+$ (so 112 $\ce{HNO3}$ to provide $112H+$ and $112NO3-$).
So the full equation:
$\ce{3C5H8 + 14K2Cr2O7 + 112HNO3 -> 15CO2 + 28Cr(NO3)3 + 28KNO3 + 68H2O}$
Let's check $\ce{NO3-}$ balance:
- In $\ce{28Cr(NO3)3}$: $28*3 = 84$ $\ce{NO3-}$
- In $\ce{28KNO3}$: $28$ $\ce{NO3-}$
- Total $\ce{NO3-}$: $84 + 28 = 112$, which matches $112HNO3$.
$\ce{K+}$ balance: $28K+$ from $14K2Cr2O7$ to $28KNO3$: balanced.
$\ce{Cr}$ balance: $28Cr$ (from $14K2Cr2O7$) to $28Cr(NO3)3$: balanced.
$\ce{C}$ balance: $15C$ (from $3C5H8$) to $15CO2$: balanced.
$\ce{H}$ balance:
- Left: $3*8H$ (from $C5H8$) + $112*1H$ (from $HNO3$) = $24 + 112 = 136H$
- Right: $68*2H$ (from $H2O$) = $136H$: balanced.
$\ce{O}$ balance:
- Left: $14*7O$ (from $K2Cr2O7$) + $112*3O$ (from $HNO3$) = $98 + 336 = 434O$
- Right: $15*2O$ (from $CO2$) + $28*9O$ (from $Cr(NO3)3$: $3*3O$ per $\ce{NO3-}$, 28 $\ce{Cr(NO3)3}$: $28*9 = 252$; $28*3O$ (from $KNO3$: $28*3 = 84$) + $68*1O$ (from $H2O$) = $30 + 252 + 84 + 68 = 434O$: balanced.
Final Balanced Equation
$\boldsymbol{\ce{3HC≡C - CH(CH3) - CH3 + 14K2Cr2O7 + 112HNO3 -> 15CO2 + 28Cr(NO3)3 + 28KNO3 + 68H2O}}$ (or using the simplified organic formula $\ce{C5H8}$: $\boldsymbol{\ce{3C5H8 + 14K2Cr2O7 + 112HNO3 -> 15CO2 + 28Cr(NO3)3 + 28KNO3 + 68H2O}}$)