QUESTION IMAGE
Question
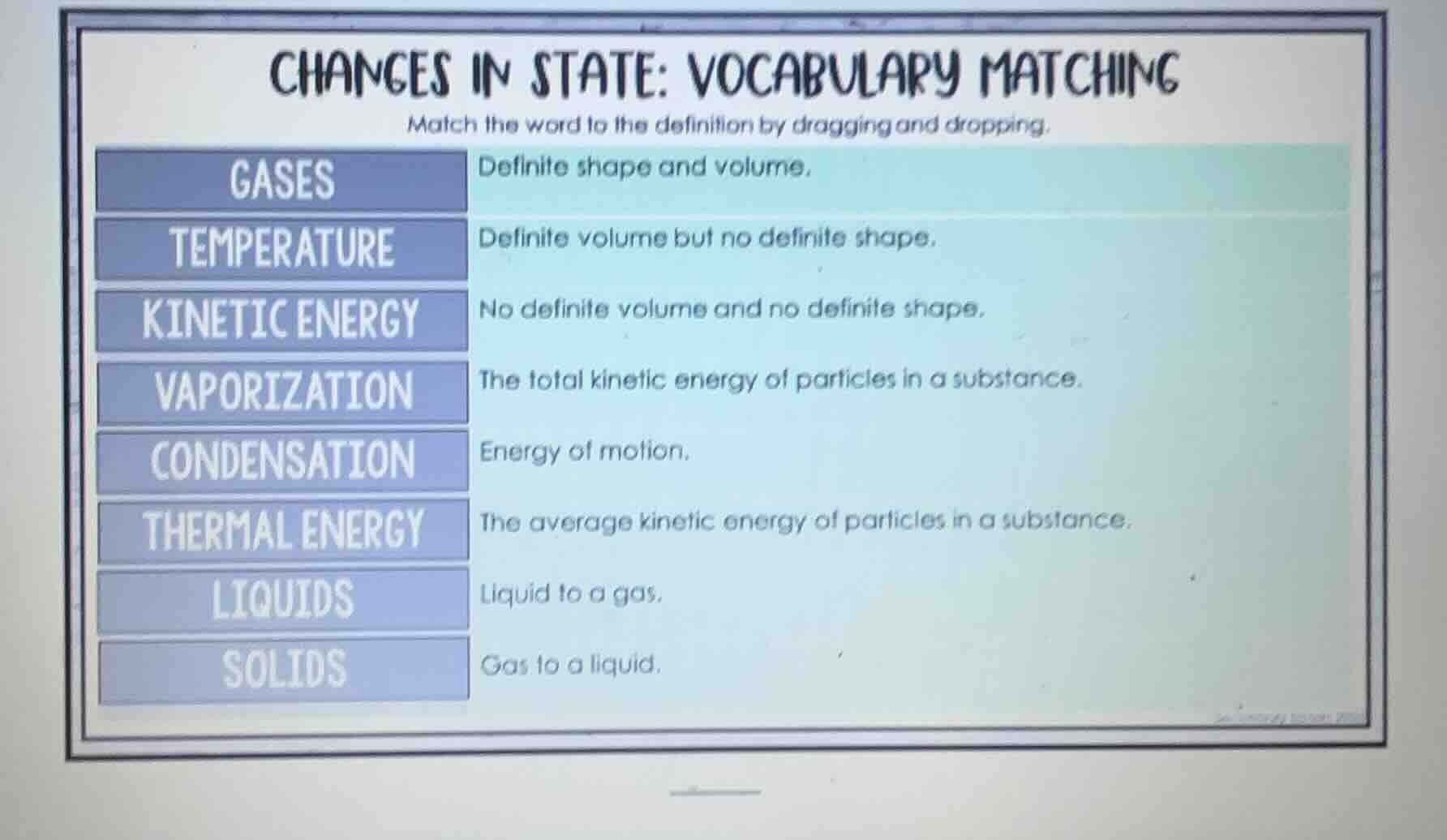
changes in state: vocabulary matching
match the word to the definition by dragging and dropping.
gases definite shape and volume.
temperature definite volume but no definite shape.
kinetic energy no definite volume and no definite shape.
vaporization the total kinetic energy of particles in a substance.
condensation energy of motion.
thermal energy the average kinetic energy of particles in a substance.
liquids liquid to a gas.
solids gas to a liquid.
To solve the vocabulary matching for changes in state, we use the definitions of each term related to states of matter and energy:
- GASES: Gases have no definite volume or shape (particles move freely).
- TEMPERATURE: Temperature is the average kinetic energy of particles in a substance.
- KINETIC ENERGY: Kinetic energy is the energy of motion (particles in motion have this).
- VAPORIZATION: Vaporization is the process of a liquid turning into a gas (e.g., boiling, evaporation).
- CONDENSATION: Condensation is the process of a gas turning into a liquid (e.g., water vapor forming dew).
- THERMAL ENERGY: Thermal energy is the total kinetic energy of all particles in a substance.
- LIQUIDS: Liquids have a definite volume (take the shape of their container, so no definite shape).
- SOLIDS: Solids have a definite shape and volume (particles are closely packed in a fixed structure).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- GASES: No definite volume and no definite shape.
- TEMPERATURE: The average kinetic energy of particles in a substance.
- KINETIC ENERGY: Energy of motion.
- VAPORIZATION: Liquid to a gas.
- CONDENSATION: Gas to a liquid.
- THERMAL ENERGY: The total kinetic energy of particles in a substance.
- LIQUIDS: Definite volume but no definite shape.
- SOLIDS: Definite shape and volume.