QUESTION IMAGE
Question
- check if each statement describes an alkali metal, an alkaline earth metal, or a transition metal.
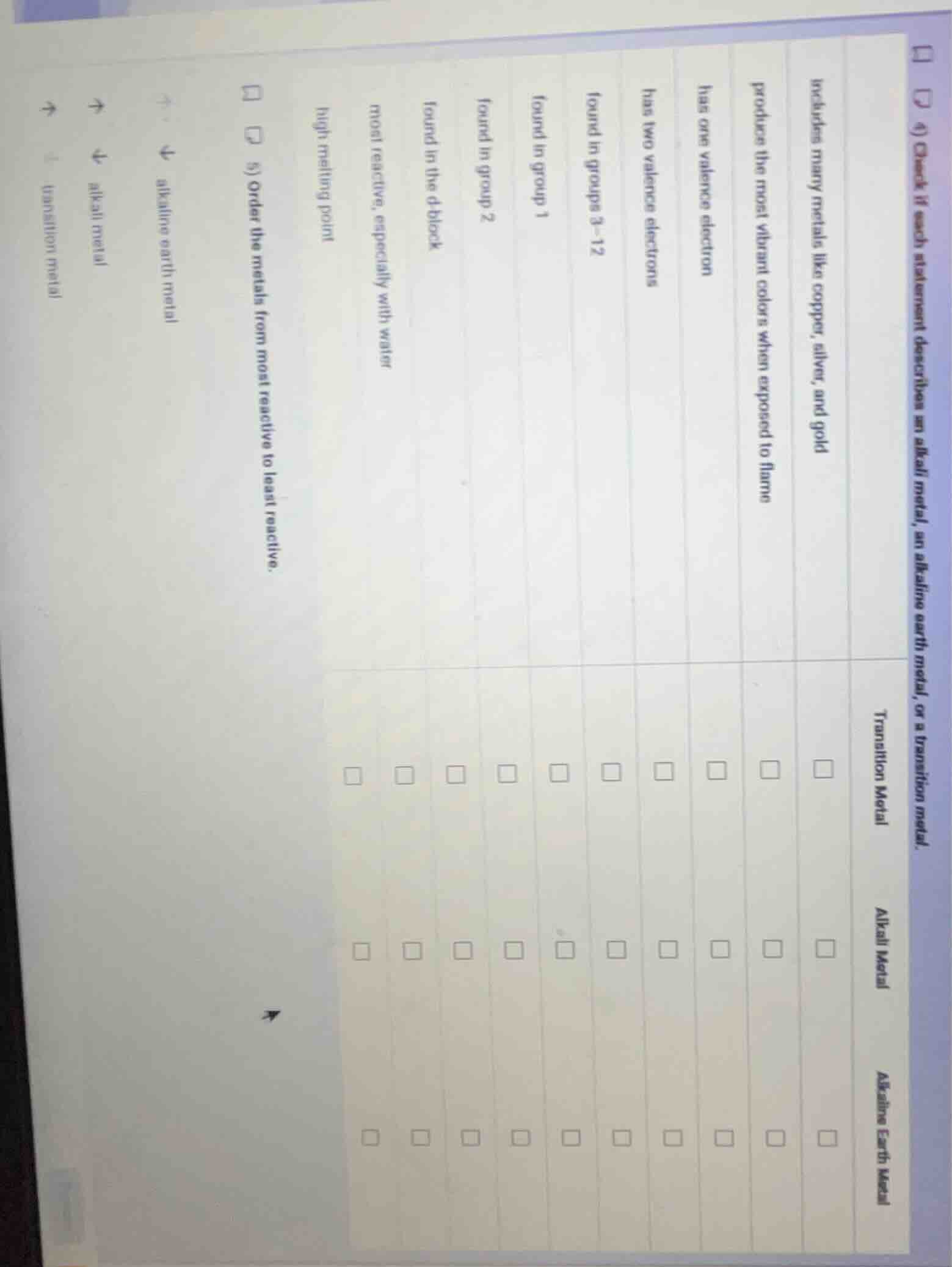
includes many metals like copper, silver, and gold
produces the most vibrant color when exposed to flame
has one valence electron
has two valence electrons
found in groups 3 - 12
found in group 1
found in group 2
found in the d block
most reactive, especially with water
high melting point
- order the metals from most reactive to least reactive.
alkaline earth metal
alkali metal
transition metal
Brief Explanations
Each statement is matched to the correct metal type based on periodic table properties and reactivity trends:
- Copper, silver, gold are classic transition metals.
- Alkali metals produce vivid flame test colors.
- Alkali metals have 1 valence electron.
- Alkaline earth metals have 2 valence electrons.
- Groups 3-12 are the d-block (transition metals).
- Group 1 contains alkali metals.
- Group 2 contains alkaline earth metals.
- The d-block corresponds to transition metals.
- Alkali metals are the most reactive with water.
- Transition metals have high melting points due to strong metallic bonding.
- Reactivity trend: Alkali metals (group 1) are most reactive, followed by alkaline earth metals (group 2), then transition metals (least reactive).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Part 4) Checkbox Matches:
| Statement | Transition Metal | Alkali Metal | Alkaline Earth Metal |
|---|---|---|---|
| produce the most vibrant colors when exposed to flame | ☐ | ☑ | ☐ |
| has one valence electron | ☐ | ☑ | ☐ |
| has two valence electrons | ☐ | ☐ | ☑ |
| found in groups 3-12 | ☑ | ☐ | ☐ |
| found in group 1 | ☐ | ☑ | ☐ |
| found in group 2 | ☐ | ☐ | ☑ |
| found in the d-block | ☑ | ☐ | ☐ |
| most reactive, especially with water | ☐ | ☑ | ☐ |
| high melting point | ☑ | ☐ | ☐ |
Part 5) Reactivity Order:
- Alkali metal
- Alkaline earth metal
- Transition metal