QUESTION IMAGE
Question
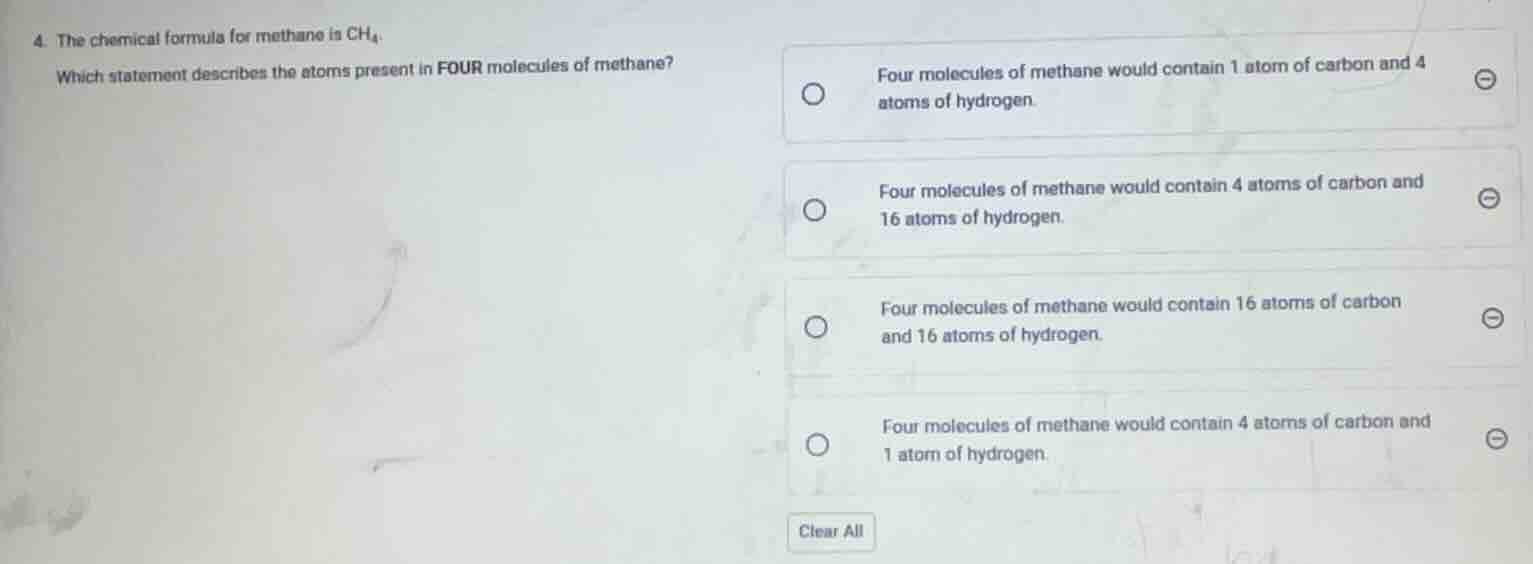
- the chemical formula for methane is $\text{ch}_4$.
which statement describes the atoms present in four molecules of methane?
four molecules of methane would contain 1 atom of carbon and 4 atoms of hydrogen.
four molecules of methane would contain 4 atoms of carbon and 16 atoms of hydrogen.
four molecules of methane would contain 16 atoms of carbon and 16 atoms of hydrogen.
four molecules of methane would contain 4 atoms of carbon and 1 atom of hydrogen.
clear all
Step1: Identify atoms per methane molecule
1 molecule of $\text{CH}_4$ has 1 C atom and 4 H atoms.
Step2: Calculate total C atoms for 4 molecules
Total C atoms = $4 \times 1 = 4$
Step3: Calculate total H atoms for 4 molecules
Total H atoms = $4 \times 4 = 16$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Four molecules of methane would contain 4 atoms of carbon and 16 atoms of hydrogen.