QUESTION IMAGE
Question
chemistry 1
ionic nomenclature
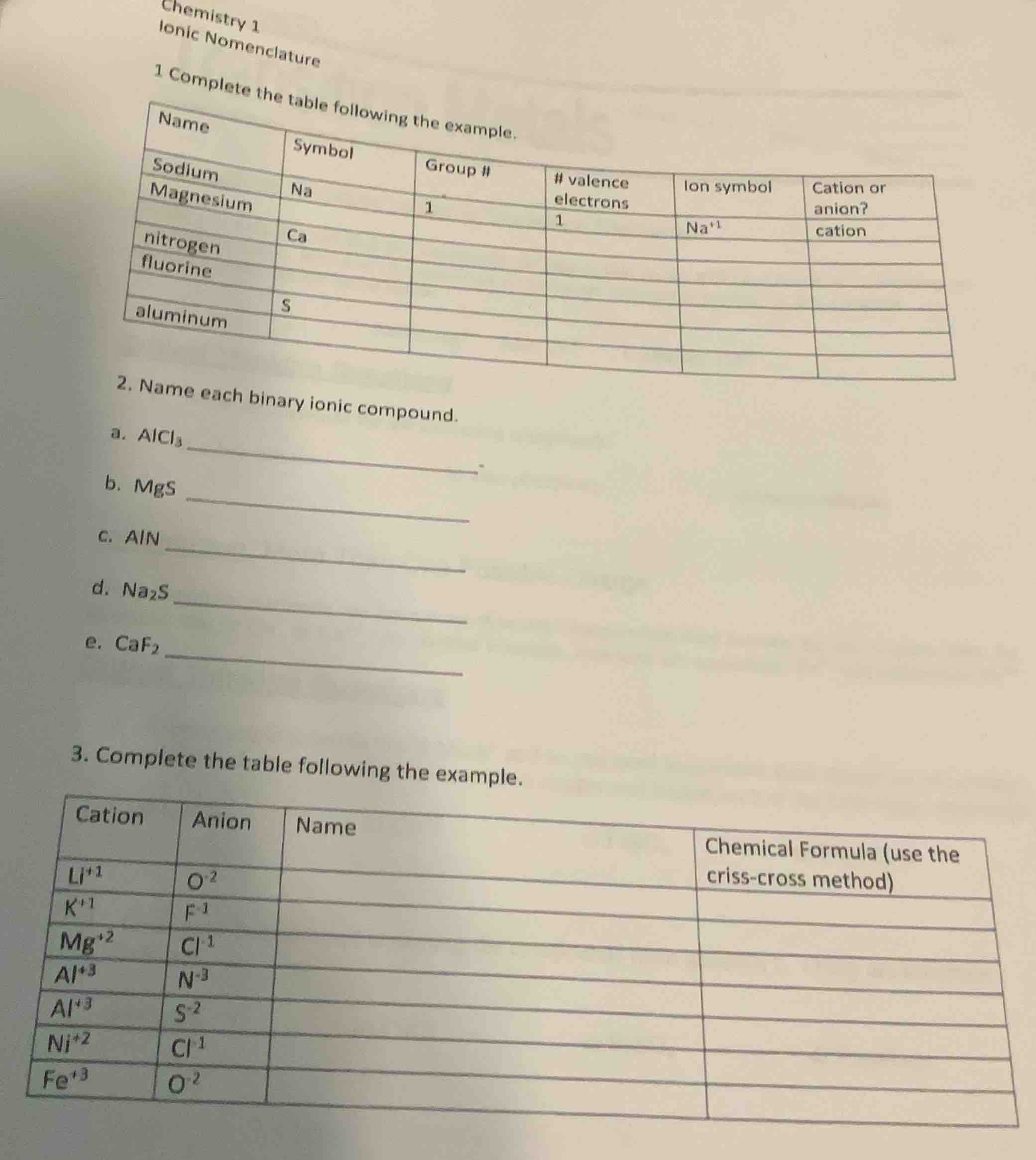
1 complete the table following the example.
name\tsymbol\tgroup #\t# valence electrons\tion symbol\tcation or anion?
sodium\tna\t1\t1\tna⁺¹\tcation
magnesium\t\t\t\t\t
\tca\t\t\t\t
nitrogen\t\t\t\t\t
fluorine\t\t\t\t\t
aluminum\t\t\t\t\t
- name each binary ionic compound.
a. alcl₃ ______.
b. mgs ______
c. aln ______
d. na₂s ______
e. caf₂ ______
- complete the table following the example.
cation\tanion\tname\tchemical formula (use the criss - cross method)
li⁺¹\to⁻²\t\t
k⁺¹\tf⁻¹\t\t
mg⁺²\tcl⁻¹\t\t
al⁺³\tn⁻³\t\t
al⁺³\ts⁻²\t\t
ni⁺²\tcl⁻¹\t\t
fe⁺³\to⁻²\t\t
1. Completed Table (Ion Properties)
| Name | Symbol | Group # | # valence electrons | Ion symbol | Cation or anion? |
|---|---|---|---|---|---|
| Magnesium | Mg | 2 | 2 | $\text{Mg}^{+2}$ | cation |
| Calcium | Ca | 2 | 2 | $\text{Ca}^{+2}$ | cation |
| Nitrogen | N | 15 | 5 | $\text{N}^{-3}$ | anion |
| Fluorine | F | 17 | 7 | $\text{F}^{-1}$ | anion |
| Sulfur | S | 16 | 6 | $\text{S}^{-2}$ | anion |
| Aluminum | Al | 13 | 3 | $\text{Al}^{+3}$ | cation |
---
2. Names of Binary Ionic Compounds
Binary ionic compounds are named by stating the cation (element name) first, then the anion (element root + "-ide"). For fixed-charge metals, no Roman numerals are needed.
Step1: Apply criss-cross method
Swap ion charges as subscripts, simplify if possible.
Step2: Name the compound
Cation name first, anion root + "-ide".
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
a. Aluminum chloride
b. Magnesium sulfide
c. Aluminum nitride
d. Sodium sulfide
e. Calcium fluoride
---