QUESTION IMAGE
Question
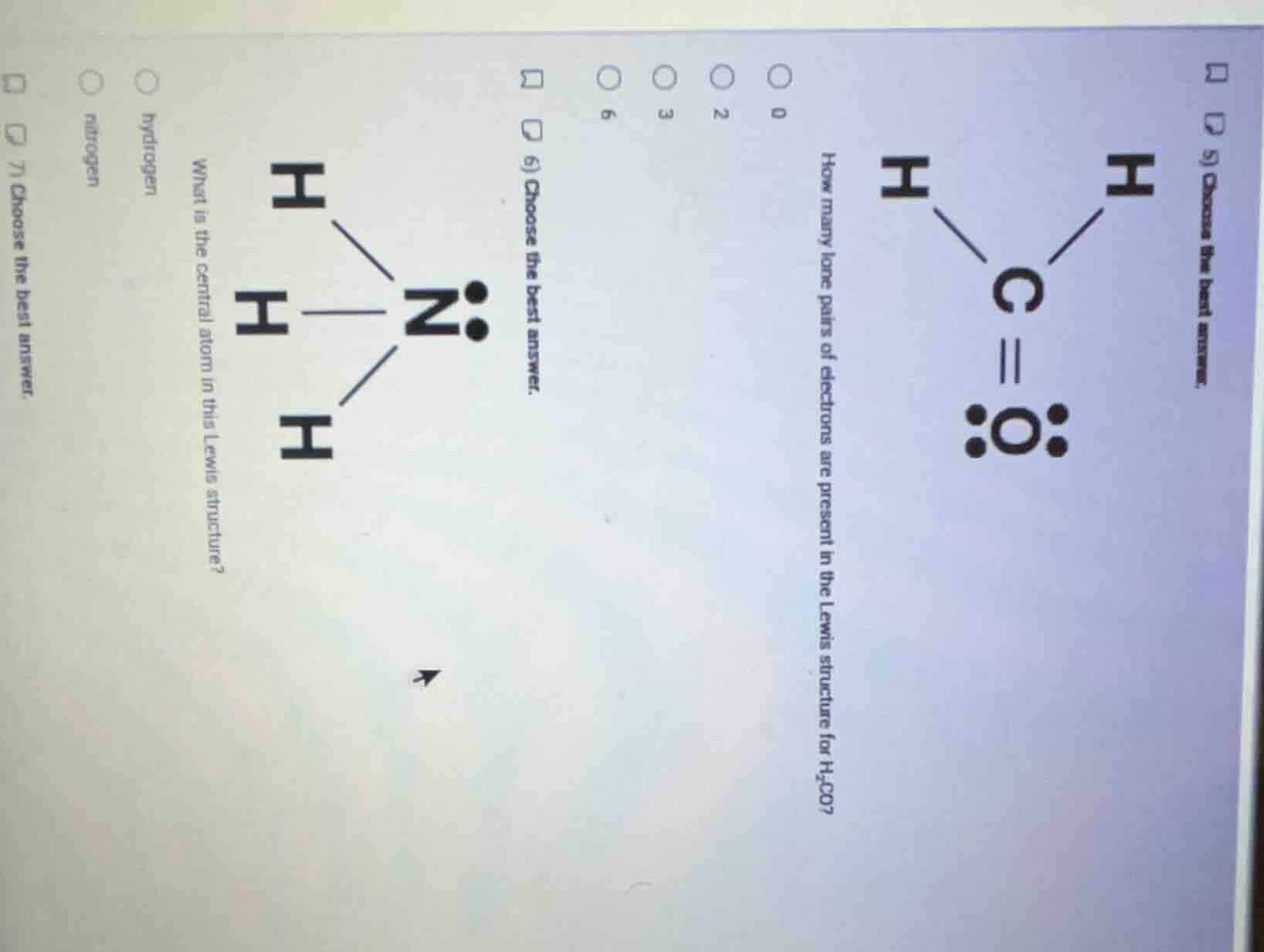
- choose the best answer. how many lone pairs of electrons are present in the lewis structure for h₂co? 0 2 3 6 6) choose the best answer. what is the central atom in this lewis structure? h h h n h hydrogen nitrogen 7) choose the best answer.
Step1: Count lone pairs in H₂CO
In the Lewis structure of $\text{H}_2\text{CO}$, only the oxygen atom has non-bonding (lone) electron pairs. Each pair is 2 electrons, and oxygen has 2 such pairs.
Step2: Identify central atom in NH₃
In the Lewis structure of $\text{NH}_3$, the nitrogen atom is bonded to 3 hydrogen atoms and has one lone pair, so it is the central atom (the atom bonded to multiple outer atoms).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- 2
- nitrogen