QUESTION IMAGE
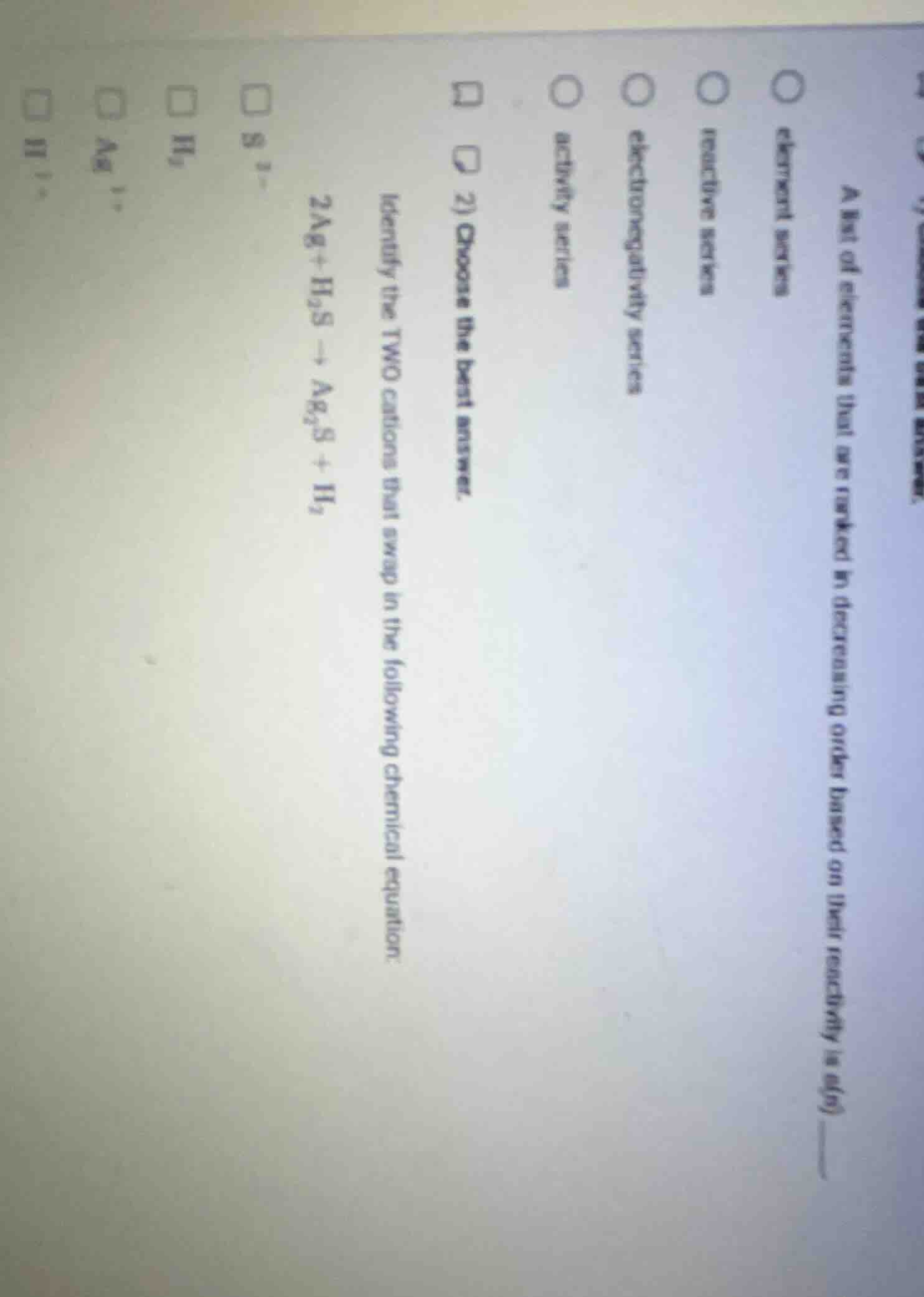
Question
- choose the best answer. identify the two cations that swap in the following chemical equation: 2ag + h₂s → ag₂s + h₂ s²⁻ h₂ ag⁺ h⁺
Step1: Analyze first question definition
A ranked list of elements by decreasing reactivity is defined as a reactive/activity series. "Reactive series" directly matches the description, while "activity series" is also a valid term, but "reactive series" is the most direct match to the wording.
Step2: Identify cations in the reaction
First, break down reactants and products into ions:
- Reactants: $2\text{Ag}$ (neutral, forms $\text{Ag}^{+}$ in product) and $\text{H}_2\text{S}$ (dissociates into $\text{H}^{+}$ and $\text{S}^{2-}$)
- Products: $\text{Ag}_2\text{S}$ (contains $\text{Ag}^{+}$ cations) and $\text{H}_2$ (neutral, formed from $\text{H}^{+}$)
The cations that swap partners are $\text{Ag}^{+}$ (from neutral Ag, bonding to $\text{S}^{2-}$) and $\text{H}^{+}$ (from $\text{H}_2\text{S}$, forming neutral $\text{H}_2$).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- reactive series
- $\text{Ag}^{+}$, $\text{H}^{+}$