QUESTION IMAGE
Question
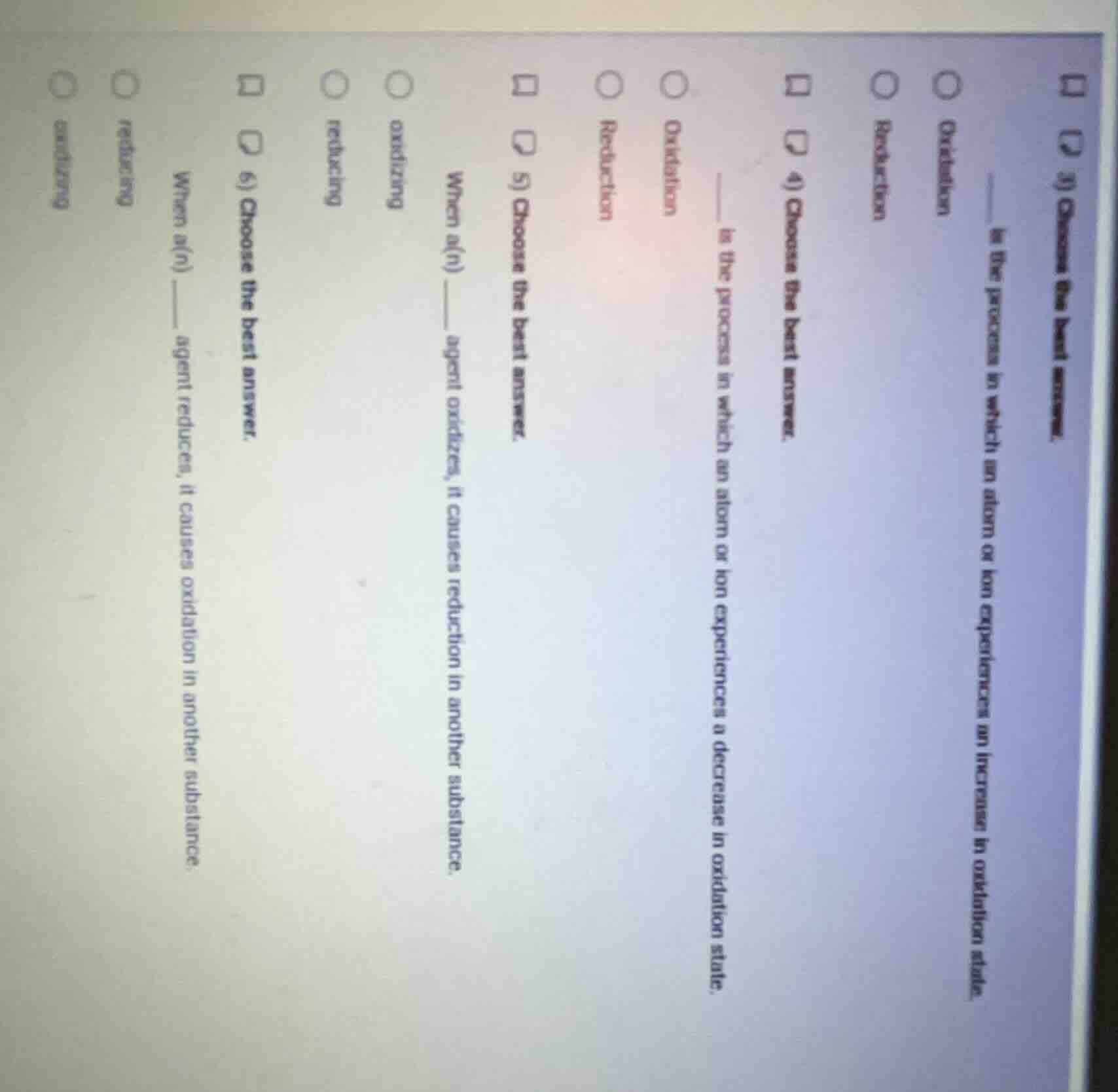
- choose the best answer. __ is the process in which an atom or ion experiences an increase in oxidation state. oxidation reduction 4) choose the best answer. is the process in which an atom or ion experiences a decrease in oxidation state. oxidation reduction 5) choose the best answer. when a(n) agent oxidizes, it causes reduction in another substance. oxidizing reducing 6) choose the best answer. when a(n) __ agent reduces, it causes oxidation in another substance. reducing oxidizing
Brief Explanations
- Oxidation is defined as the process where an atom/ion's oxidation state increases.
- Reduction is defined as the process where an atom/ion's oxidation state decreases.
- An oxidizing agent gets reduced itself, causing reduction in another substance.
- A reducing agent gets oxidized itself, causing oxidation in another substance.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Oxidation
- Reduction
- oxidizing
- reducing