QUESTION IMAGE
Question
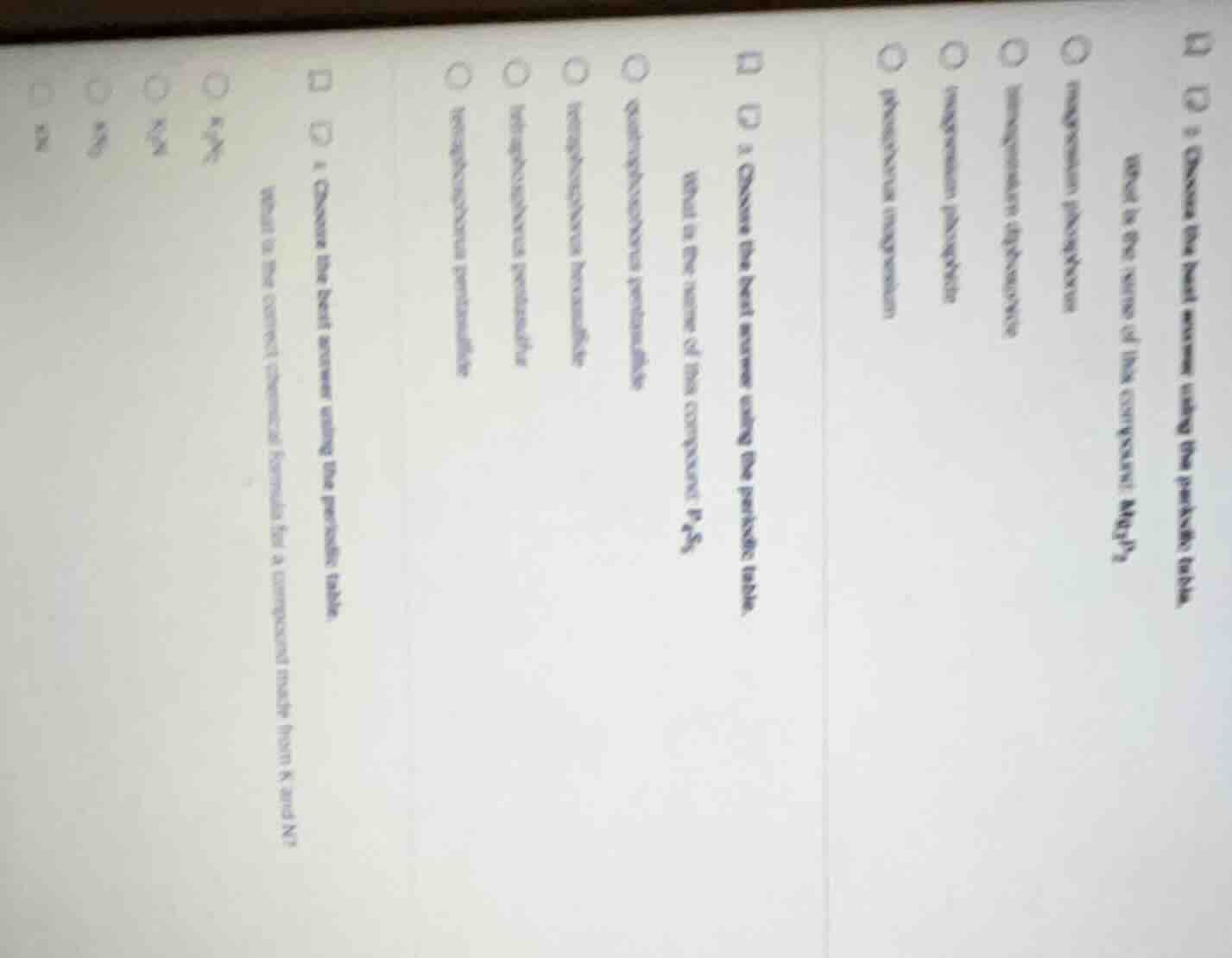
- choose the best answer using the periodic table. what is the name of this compound: p₂s₅? a) diphosphorus pentasulfide b) phosphorus pentasulfide c) phosphorus sulfide d) diphosphorus sulfide 2) choose the best answer using the periodic table. what is the correct chemical formula for a compound made from k and n? a) k₃n b) k₂n c) k₃n₂ d) kn
Step1: Name $\text{Mg}_3\text{P}_2$
Magnesium ($\text{Mg}^{2+}$) + phosphide ($\text{P}^{3-}$) gives magnesium phosphide.
Step2: Name $\text{P}_4\text{S}_5$
Tetraphosphorus ($\text{P}_4$) + pentasulfide ($\text{S}_5$) gives tetraphosphorus pentasulfide.
Step3: Formula for $\text{K}^+$ and $\text{N}^{3-}$
Balance charges: $3\text{K}^+$ + $\text{N}^{3-}$ gives $\text{K}_3\text{N}$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- magnesium phosphide
- tetraphosphorus pentasulfide
- $\text{K}_3\text{N}$