QUESTION IMAGE
Question
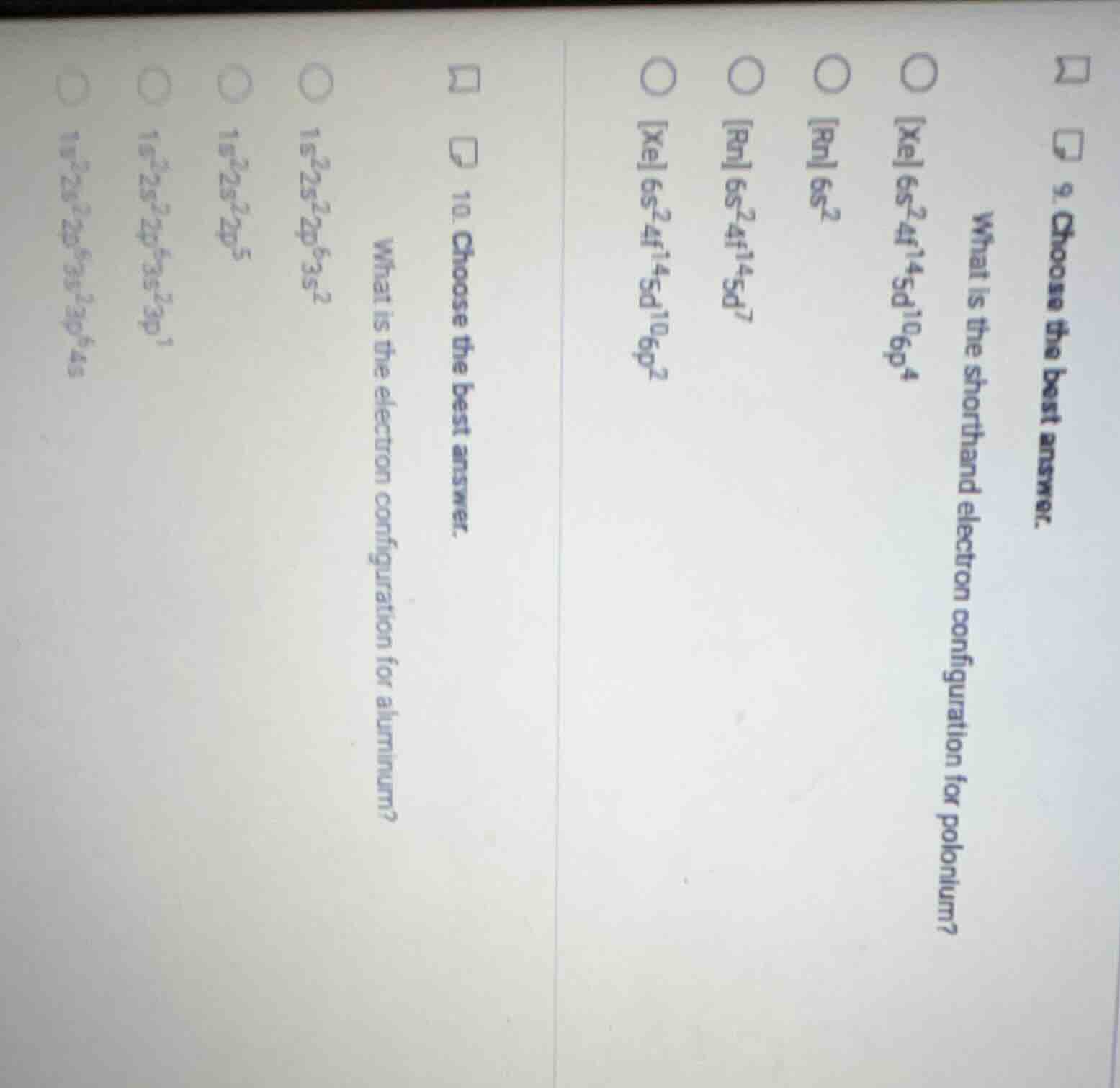
- choose the best answer. what is the shorthand electron configuration for polonium? xe 6s²4f¹⁴5d¹⁰6p⁴ rn 6s² rn 6s²4f¹⁴5d⁷ xe 6s²4f¹⁴5d¹⁰6p² 10. choose the best answer. what is the electron configuration for aluminum? 1s²2s²2p⁶3s² 1s²2s²2p⁵ 1s²2s²2p⁶3s²3p¹ 1s²2s²2p⁶3s²3p⁶4s²
Step1: Find polonium's atomic number
Polonium (Po) has an atomic number of 84.
Step2: Identify noble gas core
Xenon (Xe) is the noble gas with the highest atomic number less than 84, with an atomic number of 54.
Step3: Calculate remaining electrons
$84 - 54 = 30$ electrons to fill subshells.
Step4: Fill subshells in order
Fill $6s^2$ (2 electrons), $4f^{14}$ (14 electrons), $5d^{10}$ (10 electrons), $6p^4$ (4 electrons): $2+14+10+4=30$.
Step5: Write shorthand configuration
Combine noble gas core and valence subshells: $[Xe]6s^24f^{14}5d^{10}6p^4$
Step6: Find aluminum's atomic number
Aluminum (Al) has an atomic number of 13.
Step7: Fill subshells in order
Fill $1s^2$ (2), $2s^2$ (2), $2p^6$ (6), $3s^2$ (2), $3p^1$ (1): $2+2+6+2+1=13$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- [Xe] 6s²4f¹⁴5d¹⁰6p⁴
- 1s²2s²2p⁶3s²3p¹