QUESTION IMAGE
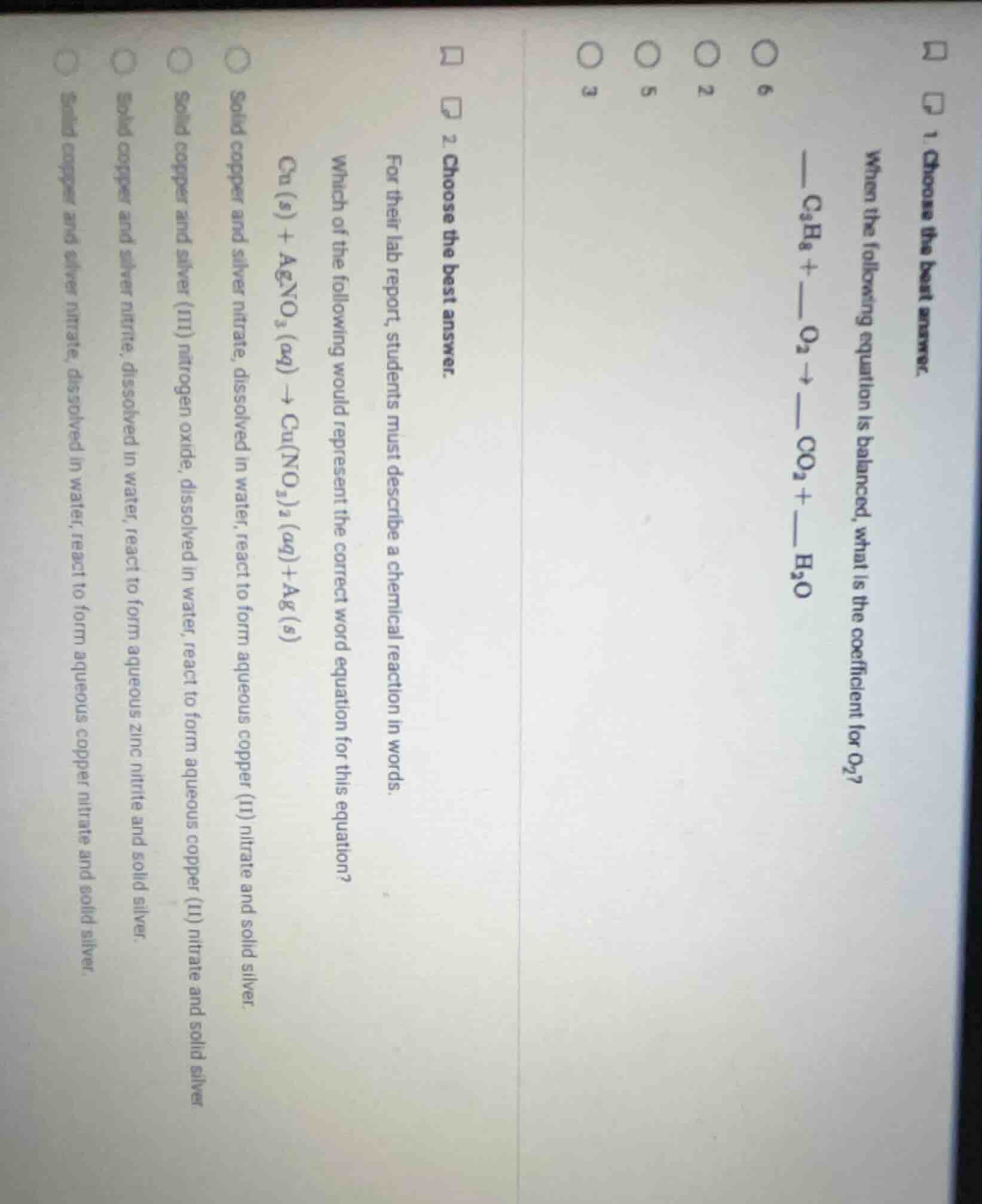
Question
- choose the best answer.
when the following equation is balanced, what is the coefficient for o₂?
——c₃h₈ + ——o₂ → ——co₂ + ——h₂o
options: 6, 2, 5, 3
- choose the best answer.
for their lab report, students must describe a chemical reaction in words.
which of the following would represent the correct word equation for this equation?
cu (s) + agno₃ (aq) → cu(no₃)₂ (aq) + ag (s)
options:
solid copper and silver nitrate, dissolved in water, react to form aqueous copper (ii) nitrate and solid silver.
solid copper and silver (iii) nitrogen oxide, dissolved in water, react to form aqueous copper (ii) nitrate and solid silver.
solid copper and silver nitrite, dissolved in water, react to form aqueous zinc nitrite and solid silver.
solid copper and silver nitrate, dissolved in water, react to form aqueous copper nitrate and solid silver.
Step1: Balance C atoms
Start with the unbalanced equation: $\ce{C3H8 + O2 -> CO2 + H2O}$.
There are 3 C on left, so set $\ce{CO2}$ coefficient to 3:
$\ce{C3H8 + O2 -> 3CO2 + H2O}$
Step2: Balance H atoms
There are 8 H on left, so set $\ce{H2O}$ coefficient to 4 (since $4\times2=8$):
$\ce{C3H8 + O2 -> 3CO2 + 4H2O}$
Step3: Balance O atoms
Count O on right: $3\times2 + 4\times1 = 10$. So set $\ce{O2}$ coefficient to 5 (since $5\times2=10$):
$\ce{C3H8 + 5O2 -> 3CO2 + 4H2O}$
Step4: Verify word equation match
Match the chemical equation $\ce{Cu(s) + AgNO3(aq) -> Cu(NO3)2(aq) + Ag(s)}$ to word descriptions:
- Reactants: Solid copper, aqueous silver nitrate (dissolved in water)
- Products: Aqueous copper(II) nitrate, solid silver
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- 5
- Solid copper and silver nitrate, dissolved in water, react to form aqueous copper (II) nitrate and solid silver.