QUESTION IMAGE
Question
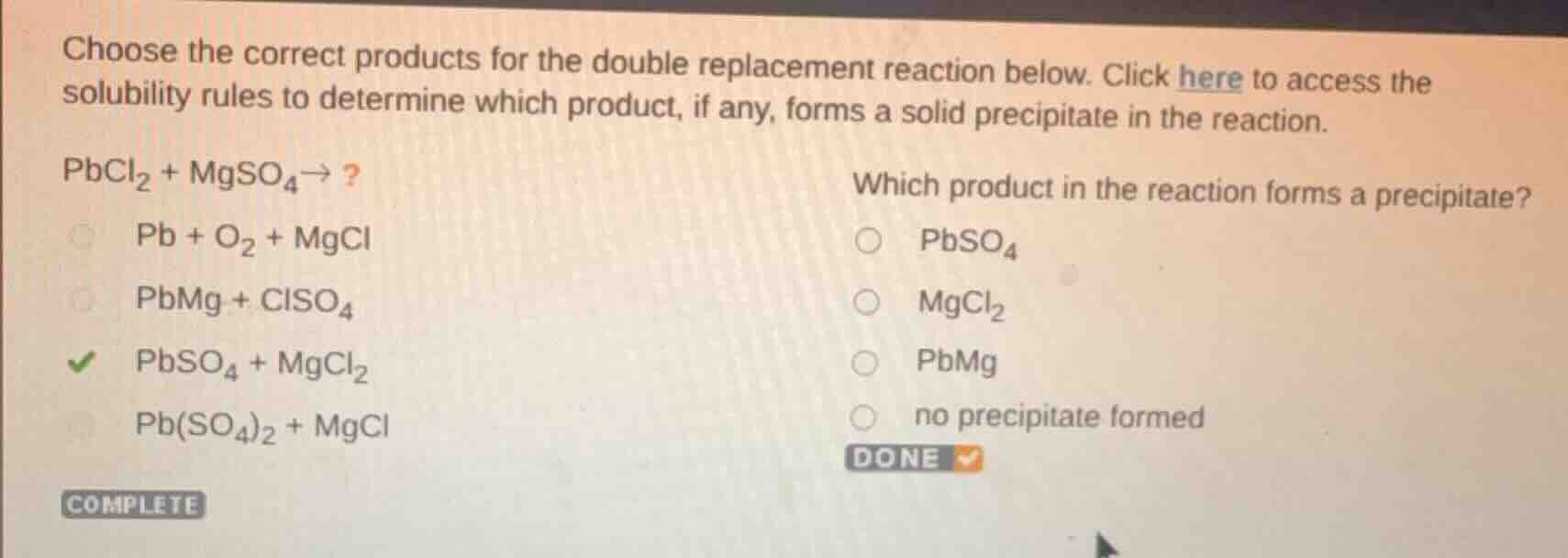
choose the correct products for the double replacement reaction below. click here to access the solubility rules to determine which product, if any, forms a solid precipitate in the reaction.\
pbcl₂ + mgso₄→?\
pb + o₂ + mgcl\
pbmg + clso₄\
pbso₄ + mgcl₂\
pb(so₄)₂ + mgcl\
which product in the reaction forms a precipitate?\
pbso₄\
mgcl₂\
pbmg\
no precipitate formed
For the first part (correct products of double replacement reaction):
Step1: Recall double replacement reaction pattern
In a double replacement reaction, the general form is \( AB + CD
ightarrow AD + CB \), where cations (\( A, C \)) and anions (\( B, D \)) swap. For \( \text{PbCl}_2 + \text{MgSO}_4 \), the cations are \( \text{Pb}^{2+} \) and \( \text{Mg}^{2+} \), anions are \( \text{Cl}^- \) and \( \text{SO}_4^{2-} \).
Step2: Apply the pattern
Swapping the cations and anions, we get \( \text{PbSO}_4 \) (from \( \text{Pb}^{2+} \) and \( \text{SO}_4^{2-} \)) and \( \text{MgCl}_2 \) (from \( \text{Mg}^{2+} \) and \( \text{Cl}^- \)). So the products are \( \text{PbSO}_4 + \text{MgCl}_2 \).
For the second part (which product forms a precipitate):
Step1: Recall solubility rules
Most sulfates are soluble, but lead(II) sulfate (\( \text{PbSO}_4 \)) is an exception and is insoluble (forms a precipitate). Magnesium chloride (\( \text{MgCl}_2 \)) is soluble (group 2 chlorides are soluble).
Step2: Determine the precipitate
From the products \( \text{PbSO}_4 \) and \( \text{MgCl}_2 \), \( \text{PbSO}_4 \) is insoluble and forms a precipitate.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Correct products: \( \text{PbSO}_4 + \text{MgCl}_2 \)
- Precipitate - forming product: \( \text{PbSO}_4 \)