QUESTION IMAGE
Question
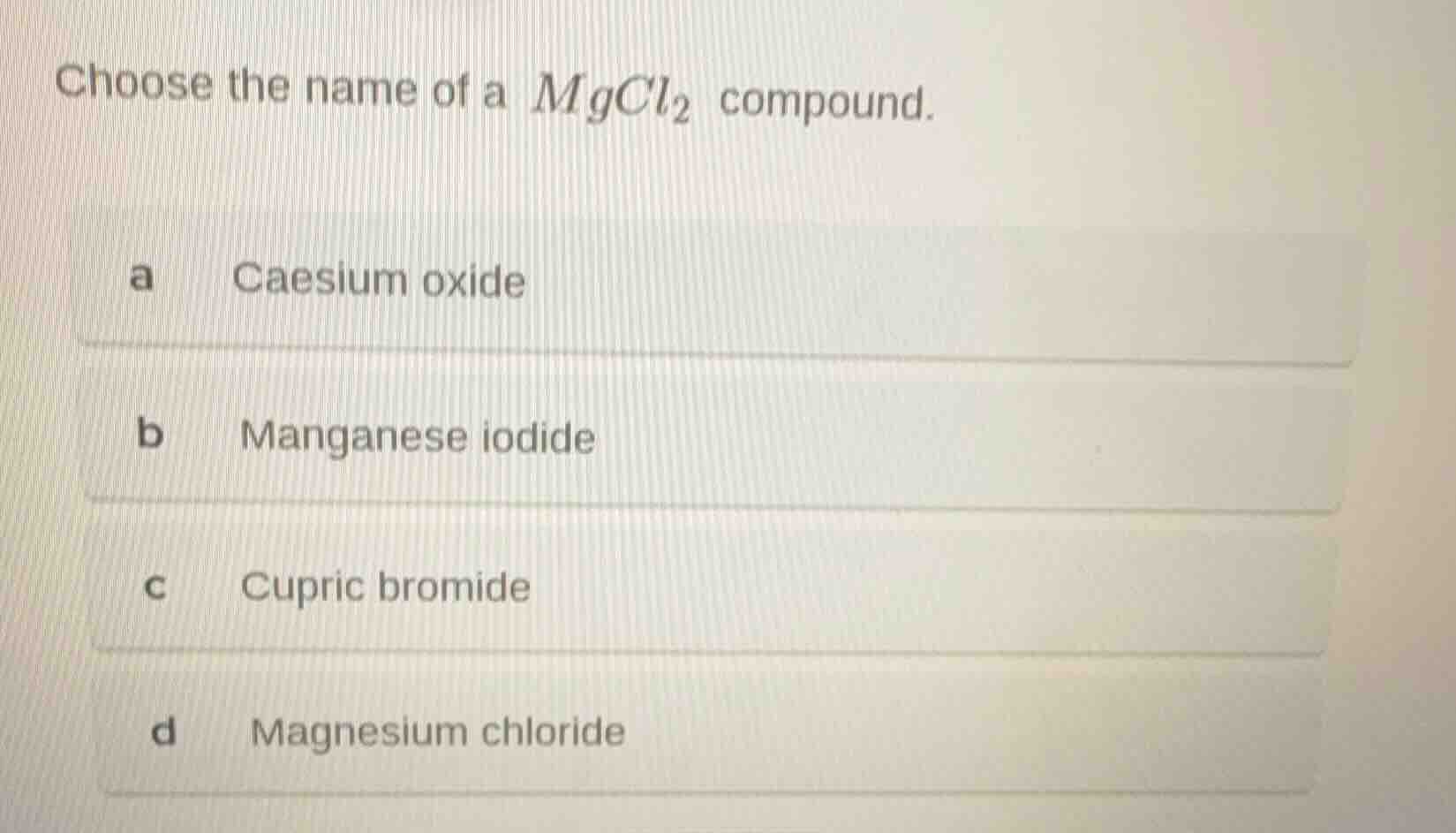
choose the name of a $mgcl_2$ compound.
a caesium oxide
b manganese iodide
c cupric bromide
d magnesium chloride
Brief Explanations
To determine the name of \( MgCl_2 \), we analyze the elements: \( Mg \) is magnesium, and \( Cl \) is chlorine (forming chloride in the compound). Option a has caesium (Cs) and oxygen (O), option b has manganese (Mn) and iodine (I), option c has copper (Cu) and bromine (Br). Only option d has magnesium (Mg) and chloride (from Cl), matching \( MgCl_2 \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
d. Magnesium chloride