QUESTION IMAGE
Question
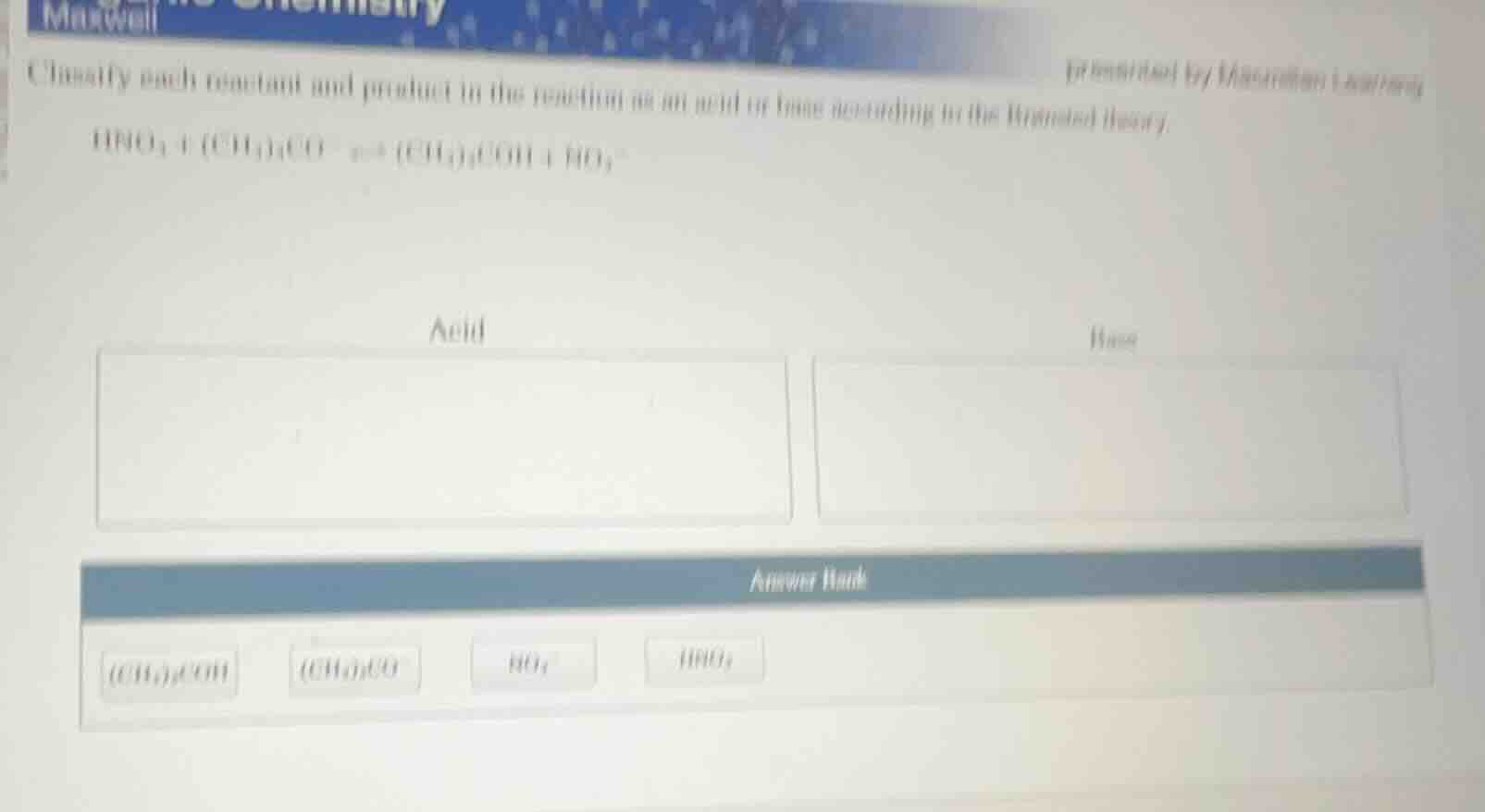
classify each reactant and product in the reaction as an acid or base according to the brønsted theory. \\( \ce{hno3 + (ch3)3co- <=> (ch3)3coh + no3-} \\)
acid
base
answer bank
\\( \ce{(ch3)3coh} \\) \\( \ce{(ch3)3co-} \\) \\( \ce{no3-} \\) \\( \ce{hno3} \\)
Brief Explanations
Per Brønsted theory, acids donate H⁺, bases accept H⁺.
- $\text{HNO}_3$ donates an H⁺ to become $\text{NO}_3^-$ → Acid.
- $\text{(CH}_3\text{)}_3\text{CO}^-$ accepts an H⁺ to become $\text{(CH}_3\text{)}_3\text{COH}$ → Base.
- $\text{(CH}_3\text{)}_3\text{COH}$ is the conjugate acid (formed when base accepts H⁺) → Acid.
- $\text{NO}_3^-$ is the conjugate base (formed when acid donates H⁺) → Base.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Acid:
$\text{HNO}_3$, $\text{(CH}_3\text{)}_3\text{COH}$
Base:
$\text{(CH}_3\text{)}_3\text{CO}^-$, $\text{NO}_3^-$