QUESTION IMAGE
Question
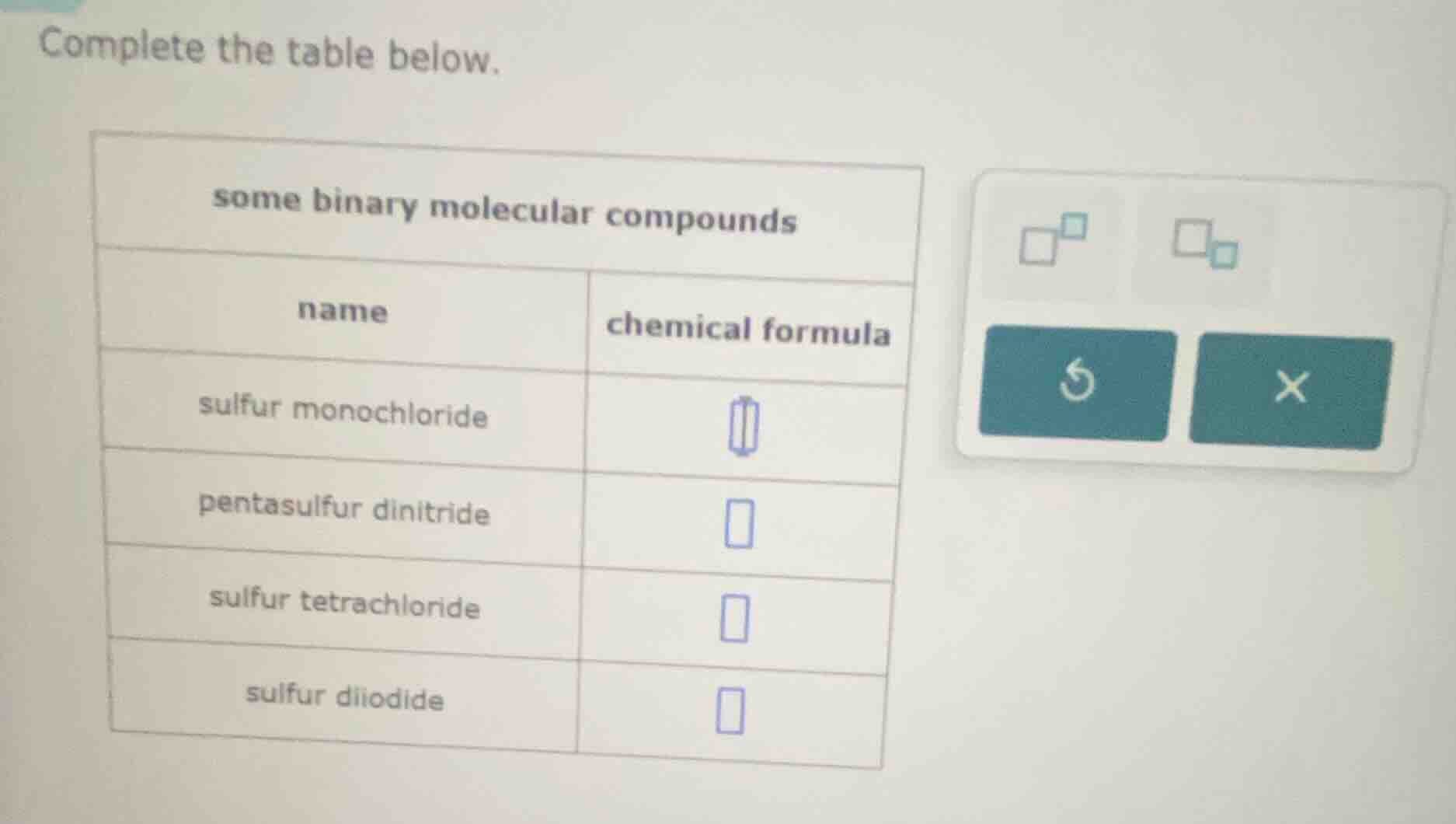
complete the table below.
some binary molecular compounds
name | chemical formula
sulfur monochloride |
pentasulfur dinitride |
sulfur tetrachloride |
sulfur diiodide |
To determine the chemical formulas of binary molecular compounds, we use the prefixes in the name to indicate the number of atoms of each element. The prefixes are: mono- (1), di- (2), tri- (3), tetra- (4), penta- (5), hexa- (6), etc. The first element in the name is usually written first in the formula, followed by the second element with its ending changed to -ide.
Step 1: Sulfur Monochloride
- The prefix "mono-" for chlorine indicates 1 chlorine atom.
- Sulfur (S) has no prefix, so it is 1 sulfur atom.
- Formula: \( SCl \)
Step 2: Pentasulfur Dinitride
- The prefix "penta-" for sulfur indicates 5 sulfur atoms.
- The prefix "di-" for nitrogen (nitride) indicates 2 nitrogen atoms.
- Formula: \( S_5N_2 \)
Step 3: Sulfur Tetrachloride
- The prefix "tetra-" for chlorine indicates 4 chlorine atoms.
- Sulfur (S) has no prefix, so it is 1 sulfur atom.
- Formula: \( SCl_4 \)
Step 4: Sulfur Diiodide
- The prefix "di-" for iodine (iodide) indicates 2 iodine atoms.
- Sulfur (S) has no prefix, so it is 1 sulfur atom.
- Formula: \( SI_2 \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Name | Chemical Formula |
|---|---|
| pentasulfur dinitride | \( S_5N_2 \) |
| sulfur tetrachloride | \( SCl_4 \) |
| sulfur diiodide | \( SI_2 \) |