QUESTION IMAGE
Question
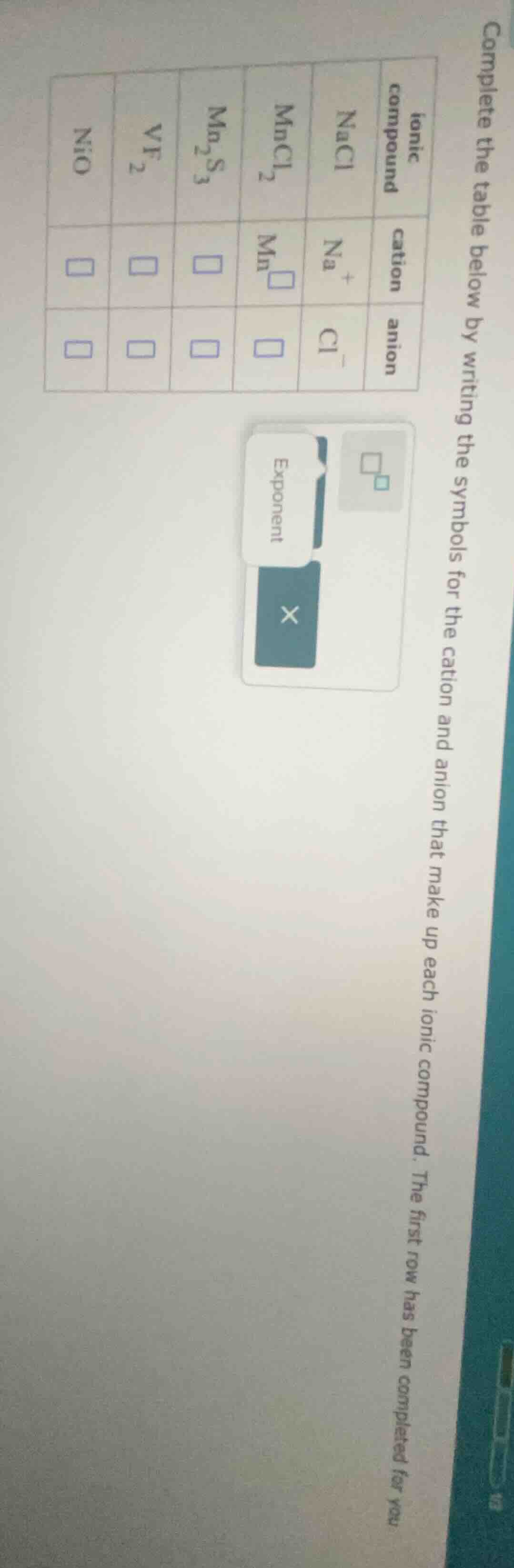
complete the table below by writing the symbols for the cation and anion that make up each ionic compound. the first row has been completed for you
| ionic compound | cation | anion |
|---|---|---|
| $ce{mncl2}$ | $ce{mn\square}$ | $square$ |
| $ce{mn2s3}$ | $square$ | $square$ |
| $ce{vf2}$ | $square$ | $square$ |
| $ce{nio}$ | $square$ | $square$ |
Step1: Analyze \( \text{MnCl}_2 \)
In \( \text{MnCl}_2 \), the anion is \( \text{Cl}^- \) (since chloride ion has a charge of \(-1\)) and to balance the charge (compound is neutral), the cation \( \text{Mn} \) must have a charge of \( +2 \) (because there are 2 \( \text{Cl}^- \) ions, so \( \text{Mn}^{2+} \) to balance \( 2\times(-1) = -2 \)). So cation: \( \text{Mn}^{2+} \), anion: \( \text{Cl}^- \).
Step2: Analyze \( \text{Mn}_2\text{S}_3 \)
For \( \text{Mn}_2\text{S}_3 \), the anion is sulfide ion \( \text{S}^{2-} \). Let the charge of \( \text{Mn} \) be \( x \). There are 2 \( \text{Mn} \) ions and 3 \( \text{S}^{2-} \) ions. So \( 2x + 3\times(-2) = 0 \Rightarrow 2x - 6 = 0 \Rightarrow x = +3 \). So cation: \( \text{Mn}^{3+} \), anion: \( \text{S}^{2-} \).
Step3: Analyze \( \text{VF}_2 \)
In \( \text{VF}_2 \), the anion is fluoride ion \( \text{F}^- \). Let the charge of \( \text{V} \) be \( y \). There is 1 \( \text{V} \) ion and 2 \( \text{F}^- \) ions. So \( y + 2\times(-1) = 0 \Rightarrow y = +2 \). So cation: \( \text{V}^{2+} \), anion: \( \text{F}^- \).
Step4: Analyze \( \text{NiO} \)
For \( \text{NiO} \), the anion is oxide ion \( \text{O}^{2-} \). Let the charge of \( \text{Ni} \) be \( z \). There is 1 \( \text{Ni} \) ion and 1 \( \text{O}^{2-} \) ion. So \( z + (-2) = 0 \Rightarrow z = +2 \). So cation: \( \text{Ni}^{2+} \), anion: \( \text{O}^{2-} \).
| Ionic Compound | Cation | Anion |
|---|---|---|
| \( \text{MnCl}_2 \) | \( \text{Mn}^{2+} \) | \( \text{Cl}^- \) |
| \( \text{Mn}_2\text{S}_3 \) | \( \text{Mn}^{3+} \) | \( \text{S}^{2-} \) |
| \( \text{VF}_2 \) | \( \text{V}^{2+} \) | \( \text{F}^- \) |
| \( \text{NiO} \) | \( \text{Ni}^{2+} \) | \( \text{O}^{2-} \) |
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The completed table is as shown above with cations and anions:
- \( \text{MnCl}_2 \): Cation \( \boldsymbol{\text{Mn}^{2+}} \), Anion \( \boldsymbol{\text{Cl}^-} \)
- \( \text{Mn}_2\text{S}_3 \): Cation \( \boldsymbol{\text{Mn}^{3+}} \), Anion \( \boldsymbol{\text{S}^{2-}} \)
- \( \text{VF}_2 \): Cation \( \boldsymbol{\text{V}^{2+}} \), Anion \( \boldsymbol{\text{F}^-} \)
- \( \text{NiO} \): Cation \( \boldsymbol{\text{Ni}^{2+}} \), Anion \( \boldsymbol{\text{O}^{2-}} \)