QUESTION IMAGE
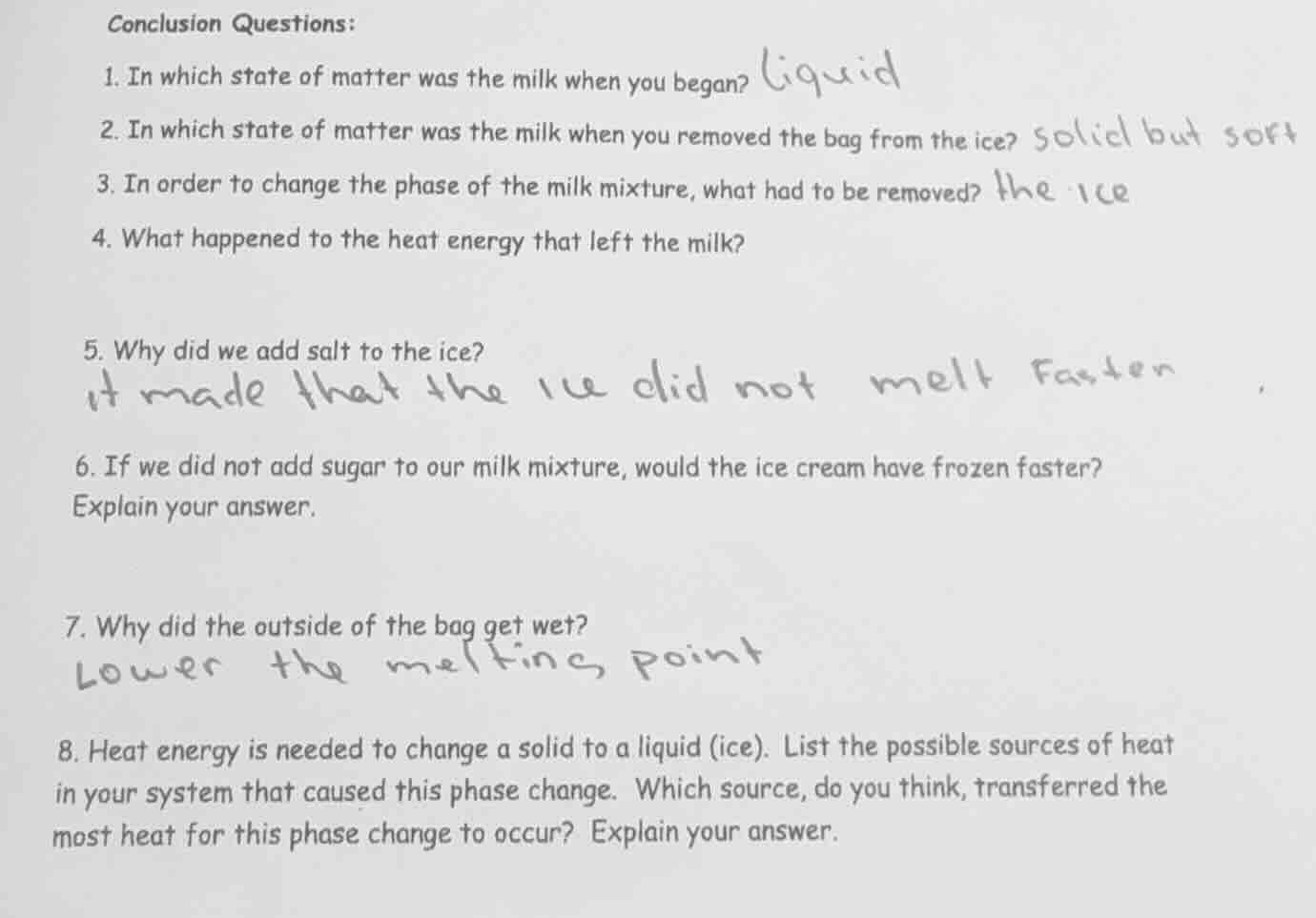
Question
conclusion questions:
- in which state of matter was the milk when you began? liquid
- in which state of matter was the milk when you removed the bag from the ice? solid but soft
- in order to change the phase of the milk mixture, what had to be removed? the ice
- what happened to the heat energy that left the milk?
- why did we add salt to the ice?
it made that the ice did not melt faster
- if we did not add sugar to our milk mixture, would the ice cream have frozen faster?
explain your answer.
- why did the outside of the bag get wet?
lower the melting point
- heat energy is needed to change a solid to a liquid (ice). list the possible sources of heat in your system that caused this phase change. which source, do you think, transferred the most heat for this phase change to occur? explain your answer.
Brief Explanations
- For question 4: Heat energy moves from warmer to cooler areas. The milk was warmer than the ice, so the heat that left the milk was absorbed by the ice and salt mixture, causing the ice to melt.
- For question 6: Sugar dissolves in milk, which lowers the freezing point of the mixture. Without sugar, the mixture's freezing point is closer to pure water's freezing point, so it would freeze faster.
- For question 7: The cold bag cools water vapor in the surrounding air, causing the water vapor to condense into liquid water droplets on the bag's surface.
- For question 8: Possible heat sources are the warm milk mixture, the surrounding air, and the hands holding the bag. The milk mixture transferred the most heat because it was the warmest component in direct contact with the ice, driving the phase change of ice to liquid as heat moved from the milk to the ice.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Question 4: The heat energy was absorbed by the ice and salt mixture, causing the ice to melt.
- Question 6: Yes, the ice cream would have frozen faster. Sugar lowers the freezing point of the milk mixture; without sugar, the mixture freezes at a higher temperature closer to pure water's freezing point, so it freezes more quickly.
- Question 7: Cold air can hold less water vapor than warm air. The cold bag cooled water vapor in the surrounding air, causing it to condense into liquid water on the bag's outside.
- Question 8:
- Possible heat sources: the warm milk mixture, surrounding air, hands holding the bag.
- The milk mixture transferred the most heat. It was the warmest substance in direct contact with the ice, so the greatest amount of heat moved from the milk to the ice to drive the solid-to-liquid phase change.