QUESTION IMAGE
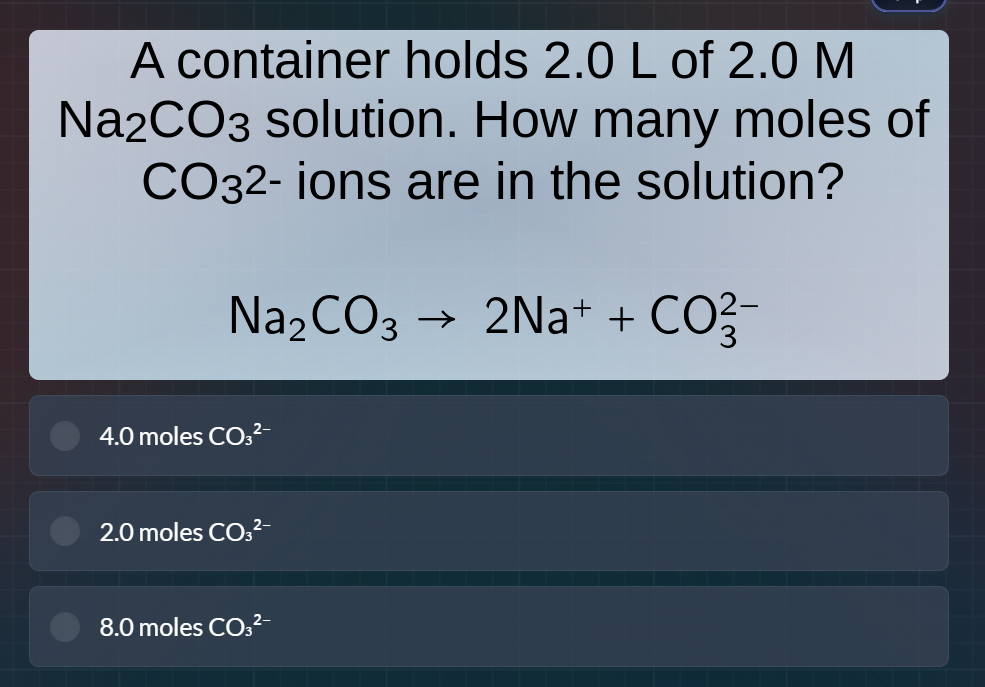
Question
a container holds 2.0 l of 2.0 m na₂co₃ solution. how many moles of co₃²⁻ ions are in the solution?
na₂co₃ → 2na⁺ + co₃²⁻
4.0 moles co₃²⁻
2.0 moles co₃²⁻
8.0 moles co₃²⁻
Step1: Calculate moles of \( Na_2CO_3 \)
Molarity formula: \( M = \frac{n}{V} \), so \( n = M \times V \).
Given \( M = 2.0 \, M \), \( V = 2.0 \, L \),
\( n(Na_2CO_3) = 2.0 \, \frac{mol}{L} \times 2.0 \, L = 4.0 \, mol \).
Step2: Relate \( Na_2CO_3 \) to \( CO_3^{2-} \)
From dissociation: \( Na_2CO_3
ightarrow 2Na^+ + CO_3^{2-} \),
1 mole \( Na_2CO_3 \) produces 1 mole \( CO_3^{2-} \).
Thus, \( n(CO_3^{2-}) = n(Na_2CO_3) = 4.0 \, mol \)? Wait, no—wait, step1: \( 2.0 M \times 2.0 L = 4.0 mol \) \( Na_2CO_3 \)? Wait, no, wait: \( 2.0 M \) is 2.0 mol/L, times 2.0 L is 4.0 mol \( Na_2CO_3 \)? But the dissociation is 1:1 for \( CO_3^{2-} \). Wait, no, wait the initial problem: wait, no, wait, maybe I made a mistake. Wait, the question is: container holds 2.0 L of 2.0 M \( Na_2CO_3 \). So moles of \( Na_2CO_3 \) is \( 2.0 M \times 2.0 L = 4.0 mol \)? But the dissociation is 1 mole \( Na_2CO_3 \) gives 1 mole \( CO_3^{2-} \). But the options have 4.0, 2.0, 8.0. Wait, no—wait, maybe I messed up. Wait, 2.0 M times 2.0 L is 4.0 mol \( Na_2CO_3 \), so 4.0 mol \( CO_3^{2-} \)? But the first option is 4.0 moles \( CO_3^{2-} \). Wait, but let's recheck.
Wait, no—wait, the molarity is 2.0 M, volume 2.0 L. Moles of \( Na_2CO_3 \) is \( M \times V = 2.0 \, mol/L \times 2.0 \, L = 4.0 \, mol \). Then, from the dissociation equation, 1 mole \( Na_2CO_3 \) produces 1 mole \( CO_3^{2-} \), so moles of \( CO_3^{2-} \) is also 4.0 mol. So the first option is 4.0 moles \( CO_3^{2-} \).
Wait, but wait, maybe I made a mistake in step1. Wait, 2.0 M is 2.0 moles per liter, times 2.0 liters is 4.0 moles of \( Na_2CO_3 \). Then, since each \( Na_2CO_3 \) dissociates into one \( CO_3^{2-} \), moles of \( CO_3^{2-} \) is 4.0 moles. So the answer is 4.0 moles \( CO_3^{2-} \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
4.0 moles \( \text{CO}_3^{2-} \) (the first option: 4.0 moles \( \text{CO}_3^{2-} \))