QUESTION IMAGE
Question
continued...
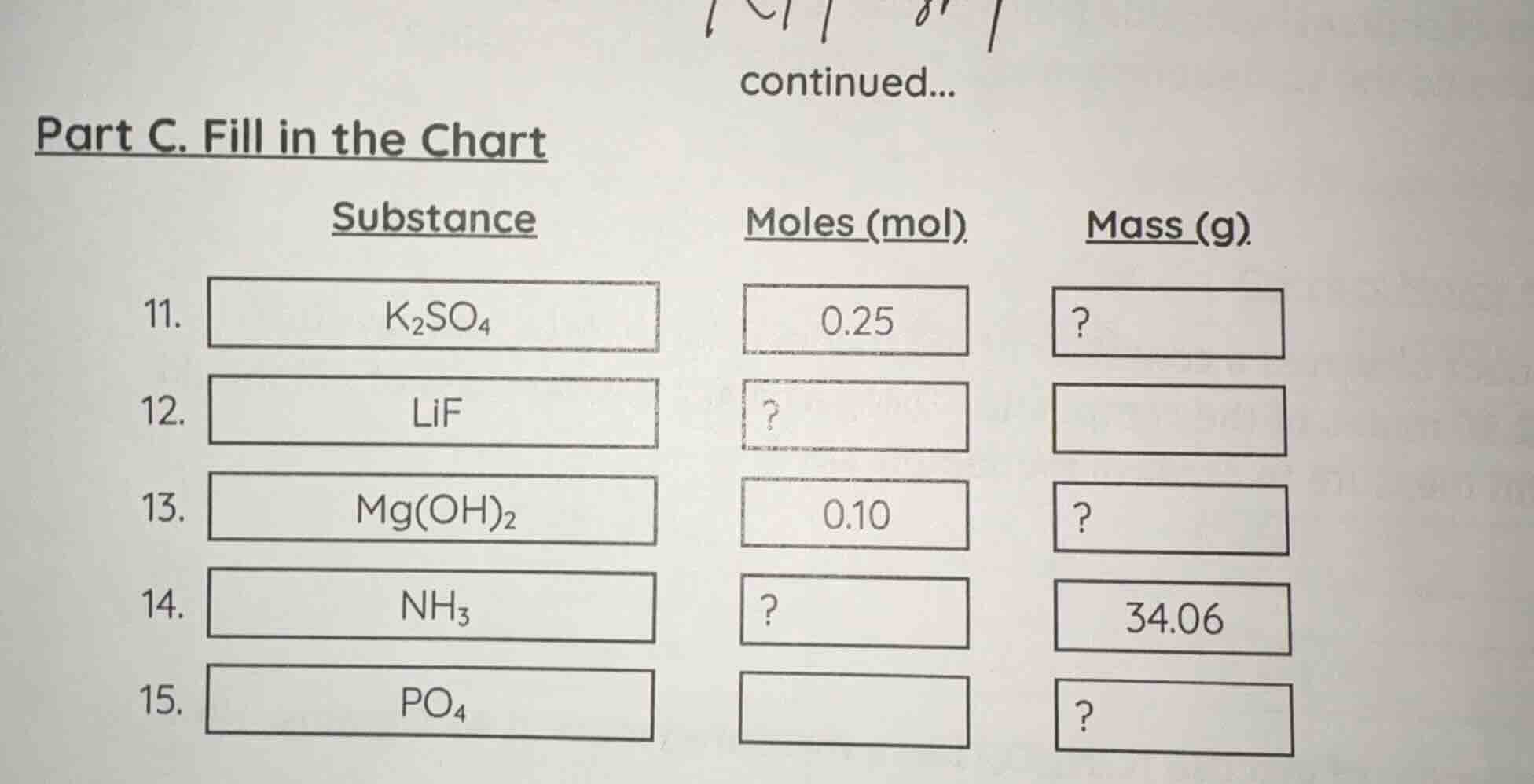
part c. fill in the chart
substance\tmoles (mol)\tmass (g)
- \tk₂so₄\t0.25\t?
- \tlif\t?\t
- \tmg(oh)₂\t0.10\t?
- \tnh₃\t?\t34.06
- \tpo₄\t\t?
Problem 11: $K_2SO_4$ with 0.25 moles, find mass
Step1: Calculate molar mass of $K_2SO_4$
Molar mass of K: $39.10\ g/mol$, S: $32.07\ g/mol$, O: $16.00\ g/mol$.
Molar mass $M = 2\times39.10 + 32.07 + 4\times16.00 = 78.20 + 32.07 + 64.00 = 174.27\ g/mol$.
Step2: Use $m = n\times M$ (mass = moles × molar mass)
$n = 0.25\ mol$, $M = 174.27\ g/mol$.
$m = 0.25\ mol\times174.27\ g/mol = 43.5675\ g \approx 43.57\ g$.
Problem 13: $Mg(OH)_2$ with 0.10 moles, find mass
Step1: Calculate molar mass of $Mg(OH)_2$
Molar mass of Mg: $24.31\ g/mol$, O: $16.00\ g/mol$, H: $1.01\ g/mol$.
Molar mass $M = 24.31 + 2\times(16.00 + 1.01) = 24.31 + 2\times17.01 = 24.31 + 34.02 = 58.33\ g/mol$.
Step2: Use $m = n\times M$
$n = 0.10\ mol$, $M = 58.33\ g/mol$.
$m = 0.10\ mol\times58.33\ g/mol = 5.833\ g \approx 5.83\ g$.
Problem 14: $NH_3$ with mass 34.06 g, find moles
Step1: Calculate molar mass of $NH_3$
Molar mass of N: $14.01\ g/mol$, H: $1.01\ g/mol$.
Molar mass $M = 14.01 + 3\times1.01 = 14.01 + 3.03 = 17.04\ g/mol$.
Step2: Use $n = \frac{m}{M}$ (moles = mass / molar mass)
$m = 34.06\ g$, $M = 17.04\ g/mol$.
$n = \frac{34.06\ g}{17.04\ g/mol} \approx 2.00\ mol$ (since $34.06 \approx 2\times17.03$, slight rounding due to molar mass precision).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
s (partial, as others need more info):
- $\boldsymbol{43.57\ g}$ (or ~43.56 g)
- $\boldsymbol{5.83\ g}$ (or ~5.83 g)
- $\boldsymbol{2.00\ mol}$ (or ~2.0 mol)
(Note: For problems 12 and 15, more info (e.g., mass for 12, moles for 15, or molar mass context) is needed. For 12, if mass was given, follow similar steps: find molar mass of LiF (Li: 6.94, F: 19.00 → $M = 25.94\ g/mol$), then $n = \frac{m}{M}$ or $m = n\times M$. For 15, $PO_4$ is a polyatomic ion (phosphate), molar mass: P: 30.97, O: 16.00 ×4 → $M = 30.97 + 64.00 = 94.97\ g/mol$, but moles are missing.)