QUESTION IMAGE
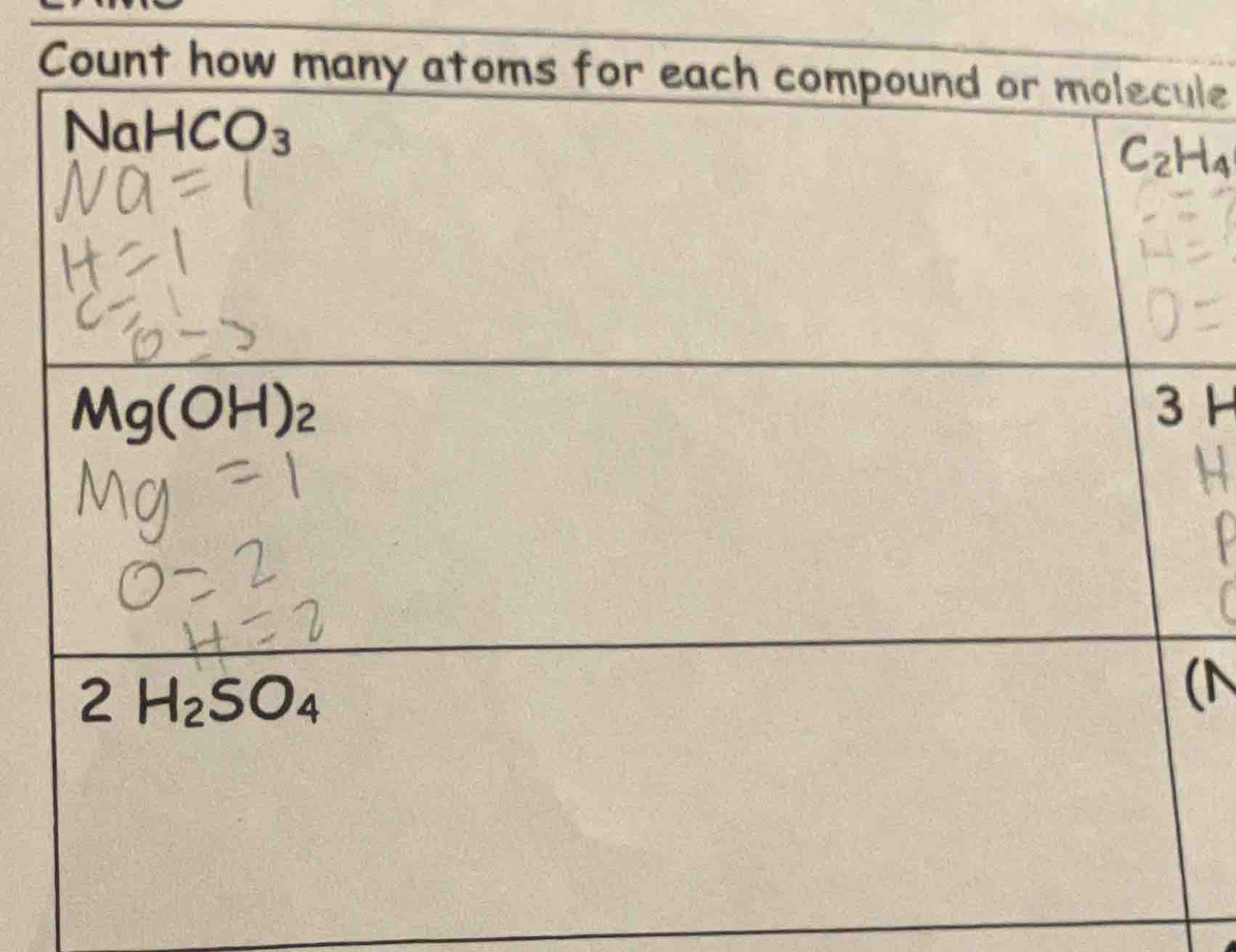
Question
count how many atoms for each compound or molecule
$ce{nahco_{3}}$
$na=1$
$h=1$
$c=1$
$o=3$
$ce{mg(oh)_{2}}$
$mg = 1$
$o=2$
$h=2$
$ce{2 h_{2}so_{4}}$
$ce{c_{2}h_{4}}$
3 h
Step1: Count atoms in $\text{NaHCO}_3$
$\text{Na}: 1, \text{H}: 1, \text{C}: 1, \text{O}: 3$; Total: $1+1+1+3=6$
Step2: Count atoms in $\text{Mg(OH)}_2$
$\text{Mg}: 1, \text{O}: 2, \text{H}: 2$; Total: $1+2+2=5$
Step3: Count atoms in $2\text{H}_2\text{SO}_4$
1 molecule: $\text{H}:2, \text{S}:1, \text{O}:4$; 2 molecules: $\text{H}:2\times2=4, \text{S}:2\times1=2, \text{O}:2\times4=8$; Total: $4+2+8=14$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\text{NaHCO}_3$: 6 total atoms (1 Na, 1 H, 1 C, 3 O)
- $\text{Mg(OH)}_2$: 5 total atoms (1 Mg, 2 O, 2 H)
- $2\text{H}_2\text{SO}_4$: 14 total atoms (4 H, 2 S, 8 O)