QUESTION IMAGE
Question
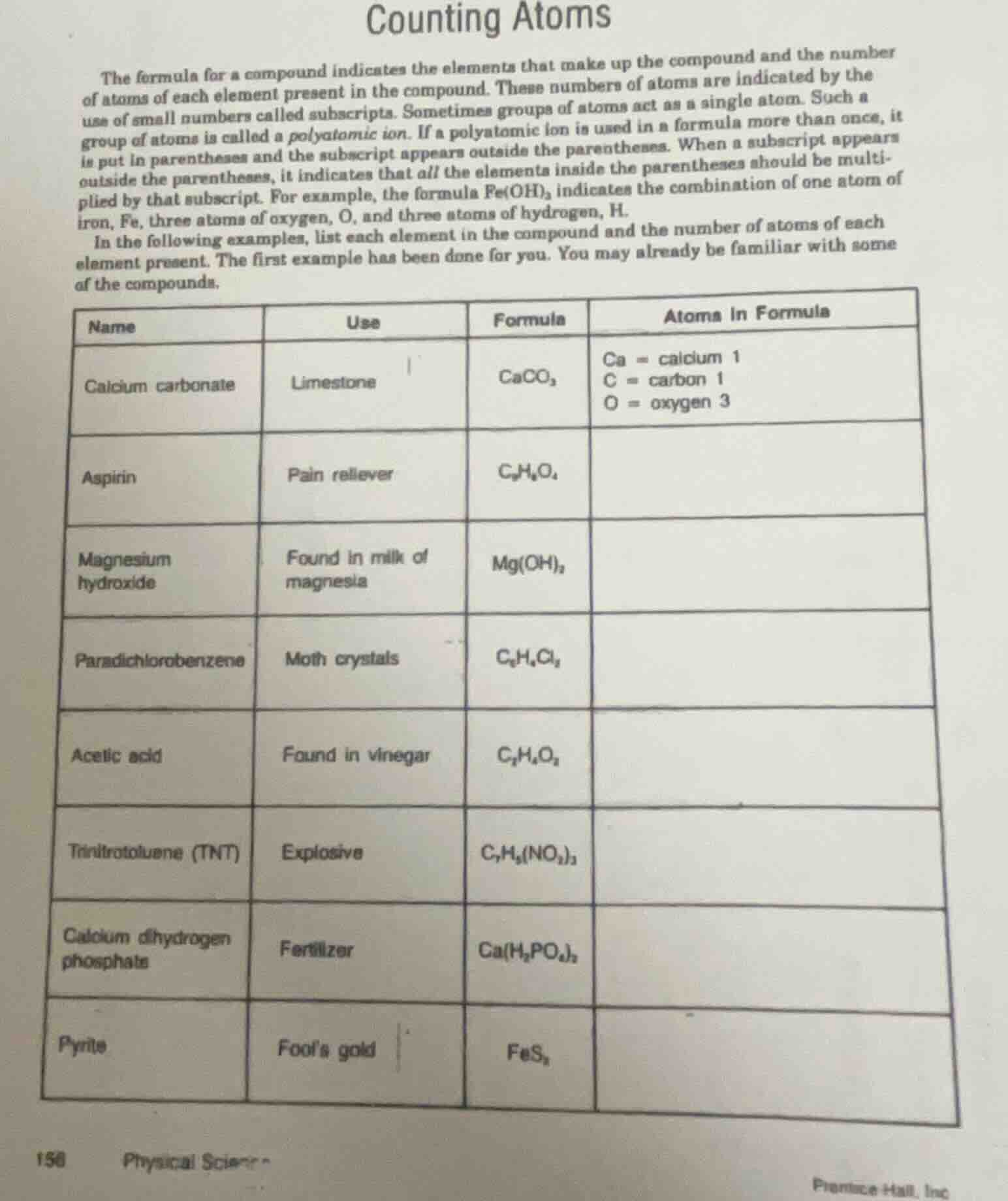
counting atoms
the formula for a compound indicates the elements that make up the compound and the number of atoms of each element present in the compound. these numbers of atoms are indicated by the use of small numbers called subscripts. sometimes groups of atoms act as a single atom. such a group of atoms is called a polyatomic ion. if a polyatomic ion is used in a formula more than once, it is put in parentheses and the subscript appears outside the parentheses. when a subscript appears outside the parentheses, it indicates that all the elements inside the parentheses should be multiplied by that subscript. for example, the formula fe(oh)₃ indicates the combination of one atom of iron, fe, three atoms of oxygen, o, and three atoms of hydrogen, h.
in the following examples, list each element in the compound and the number of atoms of each element present. the first example has been done for you. you may already be familiar with some of the compounds.
| name | use | formula | atoms in formula |
|---|---|---|---|
| aspirin | pain reliever | c₉h₈o₄ | |
| magnesium hydroxide | found in milk of magnesia | mg(oh)₂ | |
| paradichlorobenzene | moth crystals | c₆h₄cl₂ | |
| acetic acid | found in vinegar | c₂h₄o₂ | |
| trinitrotoluene (tnt) | explosive | c₇h₅(no₂)₃ | |
| calcium dihydrogen phosphate | fertilizer | ca(h₂po₄)₂ | |
| pyrite | fool’s gold | fes₂ |
Step1: Analyze aspirin formula
For $\text{C}_9\text{H}_8\text{O}_4$, subscripts give atom counts directly.
Step2: Analyze magnesium hydroxide
For $\text{Mg(OH)}_2$: Mg=1; O and H are multiplied by 2, so O=2, H=2.
Step3: Analyze paradichlorobenzene
For $\text{C}_6\text{H}_4\text{Cl}_2$, subscripts give atom counts directly.
Step4: Analyze acetic acid
For $\text{C}_2\text{H}_4\text{O}_2$, subscripts give atom counts directly.
Step5: Analyze TNT formula
For $\text{C}_7\text{H}_5(\text{NO}_2)_3$: C=7, H=5; N and O are multiplied by 3, so N=3, O=$2\times3=6$.
Step6: Analyze calcium dihydrogen phosphate
For $\text{Ca(H}_2\text{PO}_4\text{)}_2$: Ca=1; H, P, O are multiplied by 2, so H=$2\times2=4$, P=$1\times2=2$, O=$4\times2=8$.
Step7: Analyze pyrite formula
For $\text{FeS}_2$, subscripts give atom counts directly.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Aspirin: C = carbon 9, H = hydrogen 8, O = oxygen 4
- Magnesium hydroxide: Mg = magnesium 1, O = oxygen 2, H = hydrogen 2
- Paradichlorobenzene: C = carbon 6, H = hydrogen 4, Cl = chlorine 2
- Acetic acid: C = carbon 2, H = hydrogen 4, O = oxygen 2
- Trinitrotoluene (TNT): C = carbon 7, H = hydrogen 5, N = nitrogen 3, O = oxygen 6
- Calcium dihydrogen phosphates: Ca = calcium 1, H = hydrogen 4, P = phosphorus 2, O = oxygen 8
- Pyrite: Fe = iron 1, S = sulfur 2