QUESTION IMAGE
Question
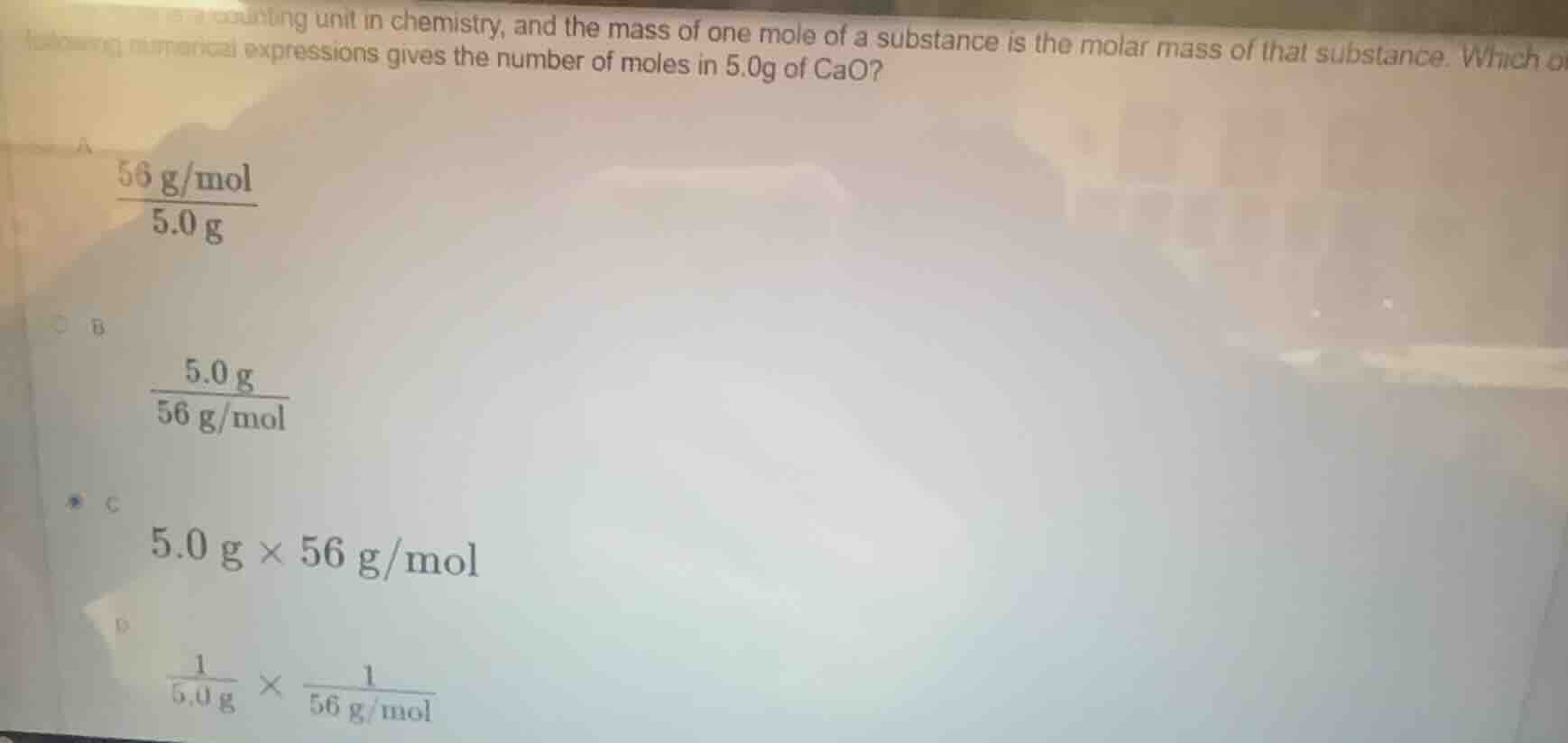
counting unit in chemistry, and the mass of one mole of a substance is the molar mass of that substance. which of the following numerical expressions gives the number of moles in 5.0g of cao? a \\(\frac{56\\ g/mol}{5.0\\ g}\\) b \\(\frac{5.0\\ g}{56\\ g/mol}\\) c \\(5.0\\ g \times 56\\ g/mol\\) d \\(\frac{1}{5.0\\ g} \times \frac{1}{56\\ g/mol}\\)
Step1: Recall mole calculation formula
The number of moles $n$ is calculated by $n = \frac{\text{mass of substance }(m)}{\text{molar mass }(M)}$
Step2: Identify values for CaO
Mass $m = 5.0\ \text{g}$, molar mass of CaO is $56\ \text{g/mol}$
Step3: Substitute values into formula
$n = \frac{5.0\ \text{g}}{56\ \text{g/mol}}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. $\frac{5.0\ \text{g}}{56\ \text{g/mol}}$