QUESTION IMAGE
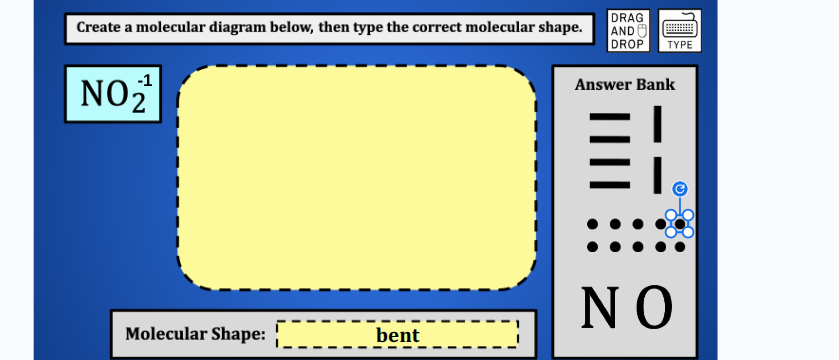
Question
create a molecular diagram below, then type the correct molecular shape.
no₂⁻¹
molecular shape: bent
answer bank
no
Step1: Identify central atom
Central atom is N.
Step2: Count valence electrons
N: 5, O: 6×2, +1 charge = $5 + 12 + 1 = 18$ total electrons.
Step3: Build bonding framework
Form N=O and N-O bonds (uses 6 electrons).
Step4: Distribute remaining electrons
Add lone pairs: 2 on double-bonded O, 3 on single-bonded O⁻, 1 lone pair on N.
Step5: Determine electron geometry
3 electron domains (2 bonds + 1 lone pair) = trigonal planar.
Step6: Determine molecular shape
1 lone pair distorts geometry to bent.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Molecular diagram: O=N-O⁻ (with one lone pair on the central N atom)
Molecular Shape: bent