QUESTION IMAGE
Question
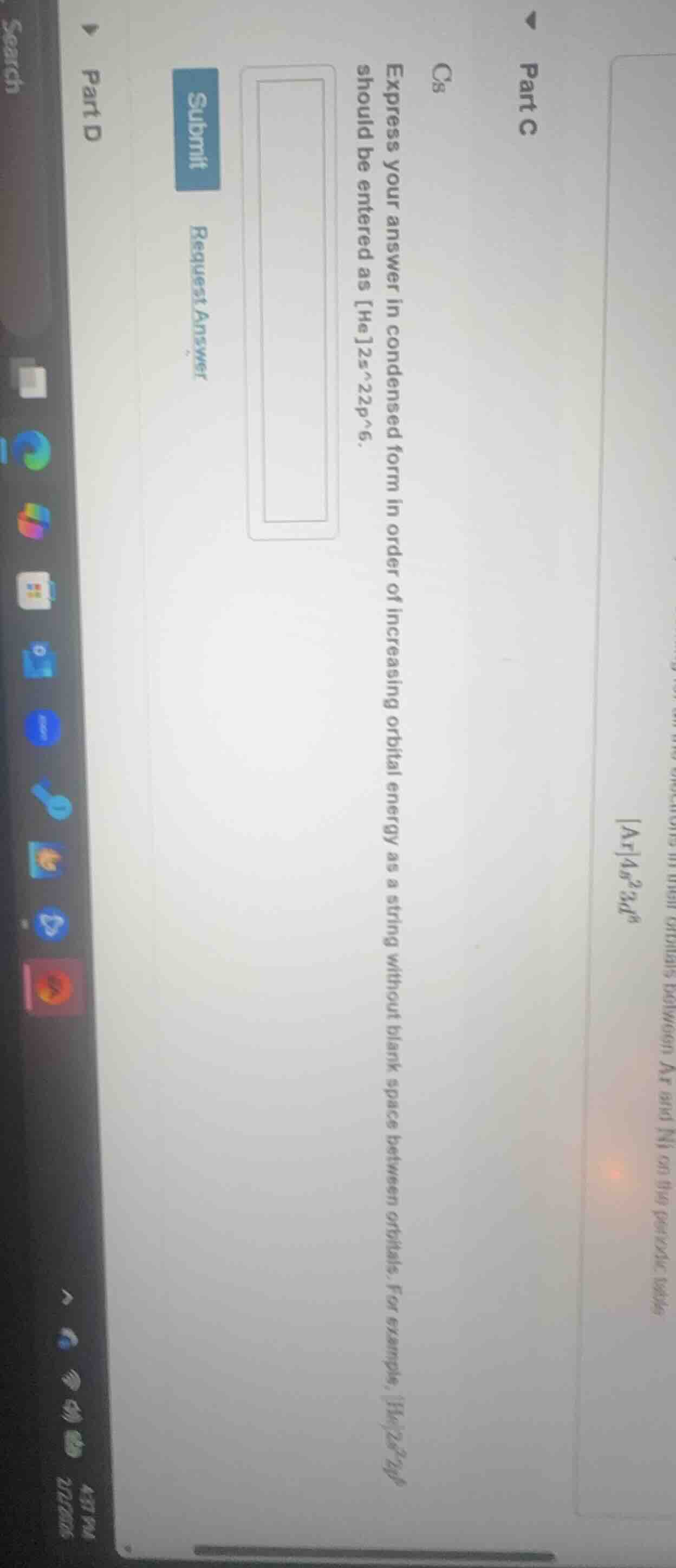
cs express your answer in condensed form in order of increasing orbital energy as a string without blank spaces between orbitals. for example, he2s^22p^6 should be entered as he2s^22p^6.
Step1: Identify noble gas core
Cesium (Cs) is in period 6, so the preceding noble gas is Xenon (Xe), with electron configuration represented as [Xe].
Step2: Add valence electrons
Cs has 1 valence electron in the 6s orbital, so we add $6s^1$ to the noble gas core.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
[Xe]6s^1