QUESTION IMAGE
Question
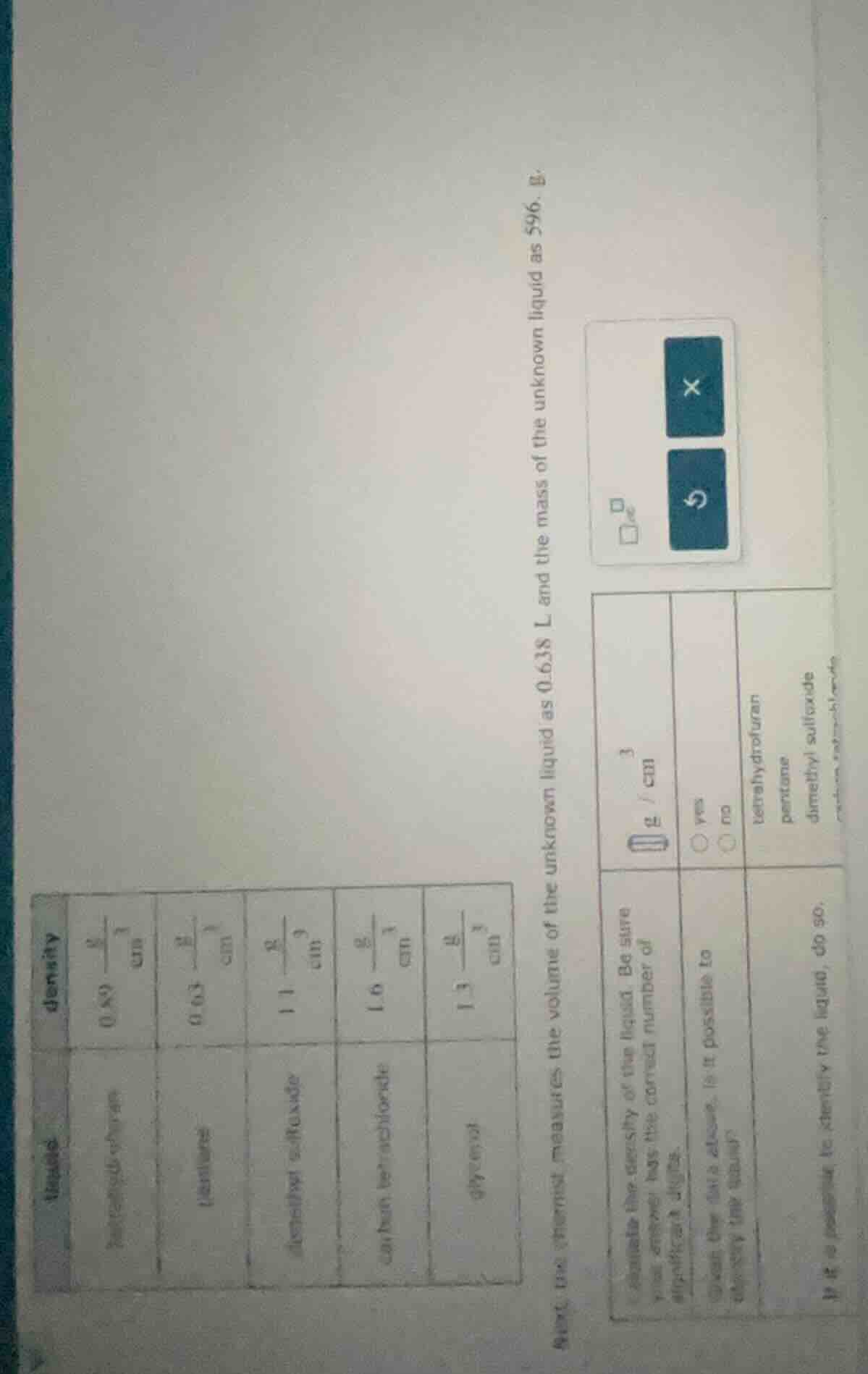
density table (partially visible) with liquids and their densities in g/cm³: some entries like 0.80, 0.03, 1.1, 1.0, 1.3. then text: an experiment measures the volume of the unknown liquid as 0.638 l and the mass as 596. b. (then a table with density unit g/cm³, yes/no option, and liquids: tetrahydrofuran, pentane, dimethyl sulfoxide... questions about calculating density, checking if possible to identify, and identifying the liquid if possible.)
Step1: Convert volume to cm³
Since $1\ \text{L} = 1000\ \text{cm}^3$,
$0.638\ \text{L} = 0.638 \times 1000 = 638\ \text{cm}^3$
Step2: Calculate density using $
ho=\frac{m}{V}$
$
ho = \frac{596\ \text{g}}{638\ \text{cm}^3} \approx 0.934\ \text{g/cm}^3$
Step3: Match density to the table
Compare the calculated density to given values; 0.93 g/cm³ matches pentane.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Density: $0.93\ \text{g/cm}^3$
Is it possible to identify the liquid? $\bigcirc$ yes
Identified liquid: pentane