QUESTION IMAGE
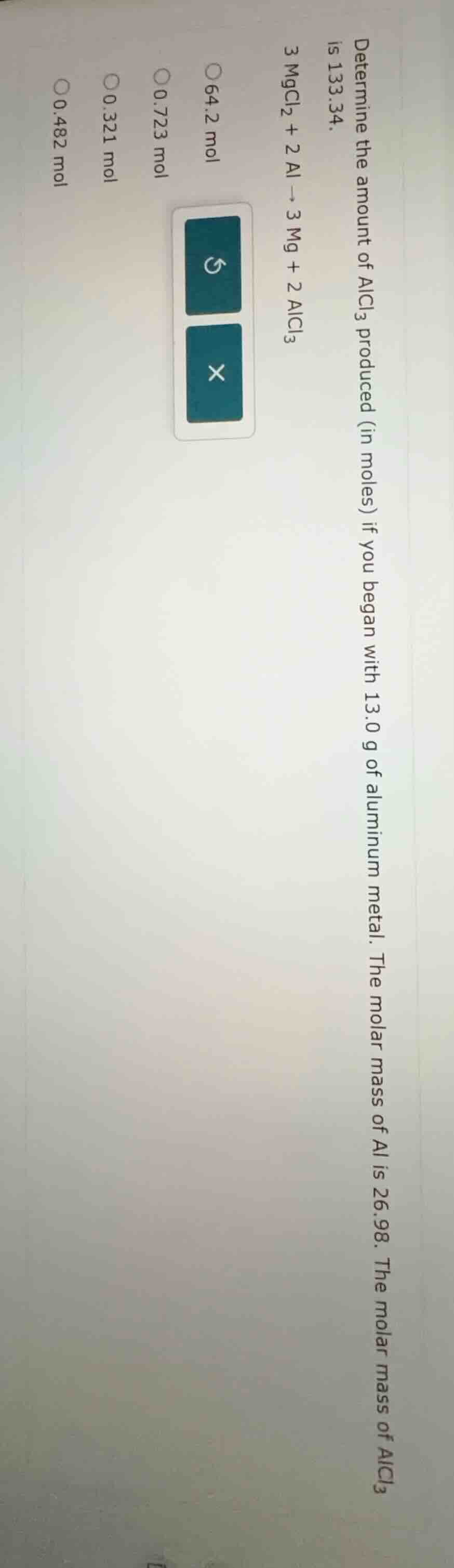
Question
determine the amount of alcl₃ produced (in moles) if you began with 13.0 g of aluminum metal. the molar mass of al is 26.98. the molar mass of alcl₃ is 133.34.
3 mgcl₂ + 2 al → 3 mg + 2 alcl₃
64.2 mol
0.723 mol
0.321 mol
0.482 mol
Step1: Calculate moles of Al
Moles of Al = $\frac{\text{mass of Al}}{\text{molar mass of Al}}$ = $\frac{13.0\ \text{g}}{26.98\ \text{g/mol}}$ ≈ 0.4818 mol
Step2: Use stoichiometry
From the reaction: 2 mol Al produces 2 mol $AlCl_3$ (mole ratio 1:1). So moles of $AlCl_3$ = moles of Al ≈ 0.482 mol
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
0.482 mol