QUESTION IMAGE
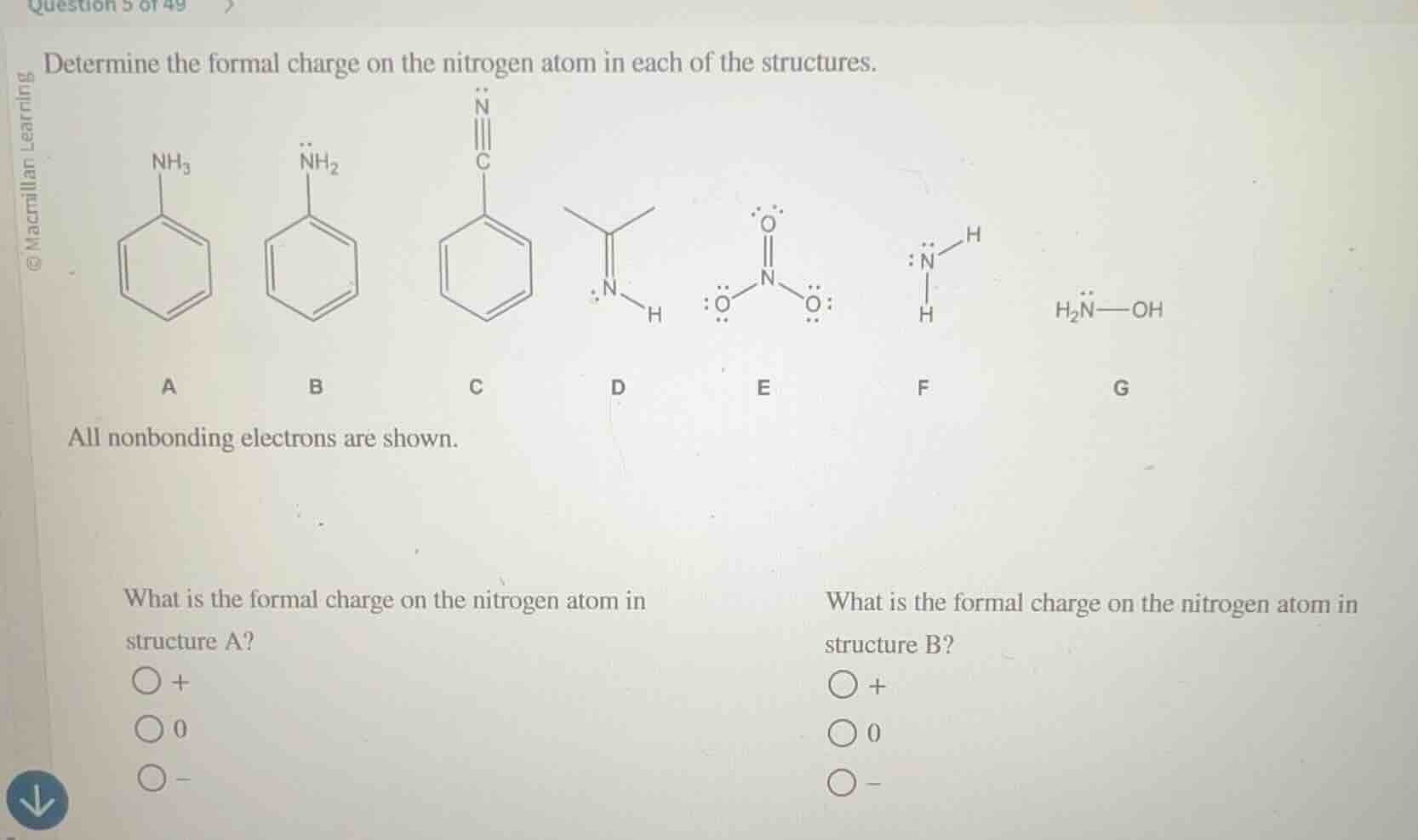
Question
determine the formal charge on the nitrogen atom in each of the structures.
all nonbonding electrons are shown.
what is the formal charge on the nitrogen atom in structure a?
+
0
-
what is the formal charge on the nitrogen atom in structure b?
+
0
-
For Structure A (NH₃ - attached to benzene ring)
Step1: Recall formal charge formula
The formula for formal charge (\(FC\)) is \(FC = V - N - \frac{B}{2}\), where \(V\) is the number of valence electrons in the neutral atom, \(N\) is the number of non - bonding electrons, and \(B\) is the number of bonding electrons. For nitrogen, \(V = 5\).
In structure A, the nitrogen atom is bonded to 3 hydrogen atoms (so \(B=6\), since each single bond has 2 electrons) and has 0 non - bonding electrons (\(N = 0\)).
Step2: Calculate formal charge
Substitute into the formula: \(FC=5 - 0-\frac{6}{2}=5 - 0 - 3 = 0\).
Step1: Recall formal charge formula
Using the same formula \(FC = V - N-\frac{B}{2}\), with \(V = 5\) for nitrogen.
In structure B, the nitrogen atom is bonded to 2 hydrogen atoms (so \(B = 4\), as each single bond has 2 electrons) and has 2 non - bonding electrons (\(N=2\)).
Step2: Calculate formal charge
Substitute into the formula: \(FC = 5-2-\frac{4}{2}=5 - 2-2 = 0\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
0 (corresponding to the option "0")