QUESTION IMAGE
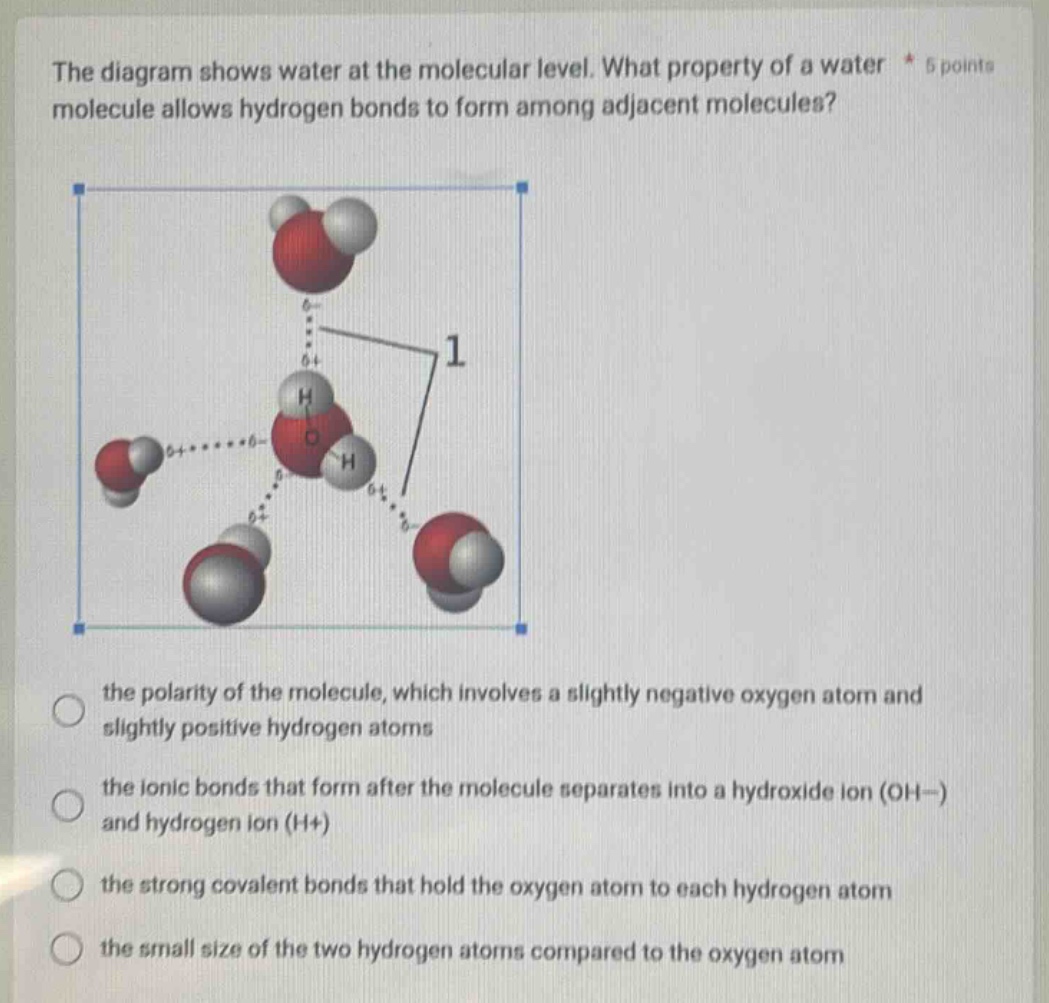
Question
the diagram shows water at the molecular level. what property of a water molecule allows hydrogen bonds to form among adjacent molecules? the polarity of the molecule, which involves a slightly negative oxygen atom and slightly positive hydrogen atoms the ionic bonds that form after the molecule separates into a hydroxide ion (oh−) and hydrogen ion (h+) the strong covalent bonds that hold the oxygen atom to each hydrogen atom the small size of the two hydrogen atoms compared to the oxygen atom
To determine the property of a water molecule that allows hydrogen bonds to form among adjacent molecules, we analyze each option:
- Option 1 (the polarity of the molecule, which involves a slightly negative oxygen atom and slightly positive hydrogen atoms): Water is a polar molecule because oxygen is more electronegative than hydrogen. This causes the oxygen atom to have a partial negative charge ($\delta^-$) and the hydrogen atoms to have partial positive charges ($\delta^+$). The partial positive charge of a hydrogen atom in one water molecule is attracted to the partial negative charge of the oxygen atom in an adjacent water molecule, forming a hydrogen bond. This matches the mechanism of hydrogen bond formation in water.
- Option 2 (the ionic bonds that form after the molecule separates into a hydroxide ion (OH⁻) and hydrogen ion (H⁺)): Ionic bonds are not involved in hydrogen bond formation between water molecules. Water molecules form hydrogen bonds while they are still intact (as H₂O molecules), not when they dissociate into ions. So this option is incorrect.
- Option 3 (the strong covalent bonds that hold the oxygen atom to each hydrogen atom): Covalent bonds hold the atoms within a single water molecule together (intramolecular bonds), but hydrogen bonds are intermolecular forces between different water molecules. The strength of the intramolecular covalent bonds does not directly relate to the formation of intermolecular hydrogen bonds. Thus, this option is incorrect.
- Option 4 (the small size of the two hydrogen atoms compared to the oxygen atom): The size of the atoms does not determine the ability to form hydrogen bonds. Hydrogen bonding is based on the polarity of the molecule and the resulting partial charges, not the relative sizes of the atoms. So this option is incorrect.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
the polarity of the molecule, which involves a slightly negative oxygen atom and slightly positive hydrogen atoms