QUESTION IMAGE
Question
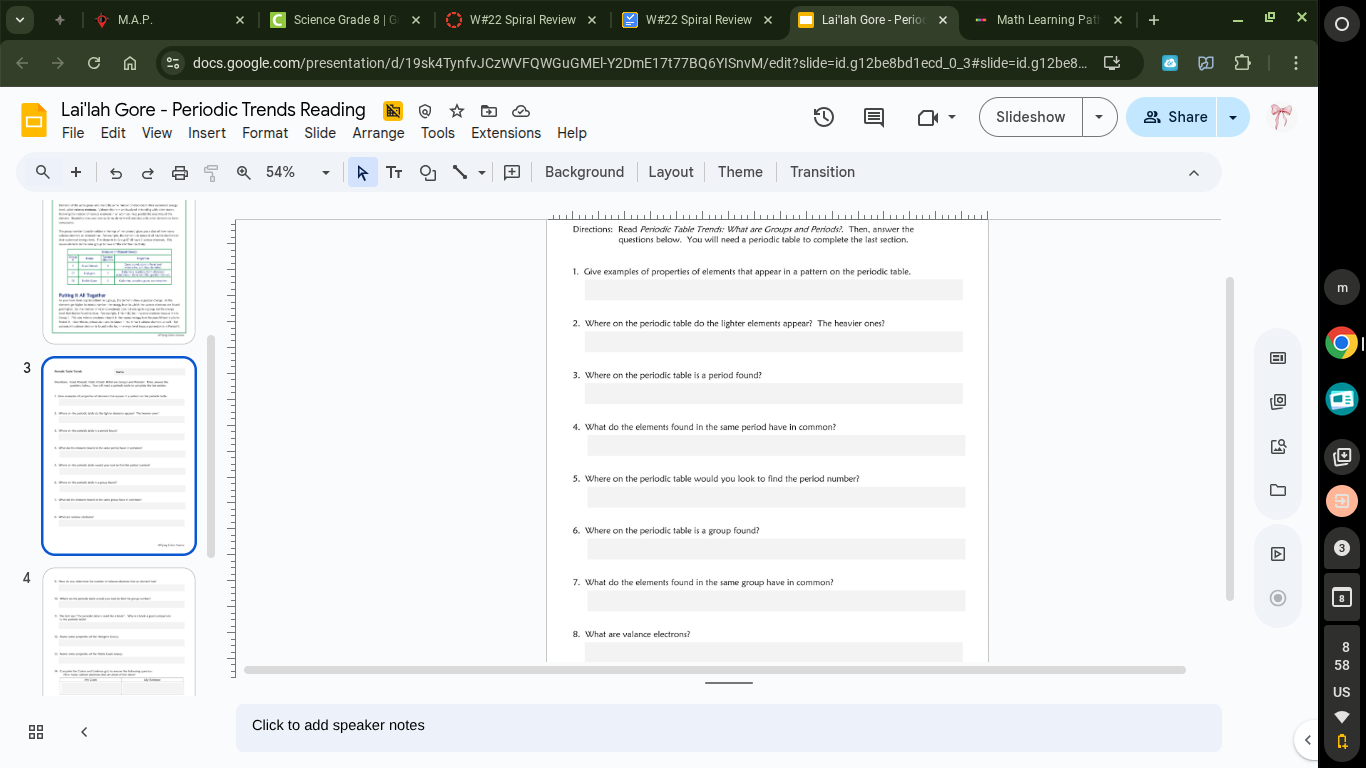
directions: read periodic table trends: what are groups and periods? then, answer the questions below. you will need a periodic table to complete the last question. 1. give examples of properties of elements that appear in a pattern on the periodic table. 2. where on the periodic table do the lighter elements appear? the heavier ones? 3. where on the periodic table is a period found? 4. what do the elements found in the same period have in common? 5. where on the periodic table would you look to find the period number? 6. where on the periodic table is a group found? 7. what do the elements found in the same group have in common? 8. what are valence electrons?
To answer these periodic table trend questions, we analyze each one based on chemistry concepts:
1. Properties of elements in a pattern
Elements show patterns in atomic radius (decreases across a period, increases down a group), ionization energy (increases across a period, decreases down a group), electronegativity (increases across a period, decreases down a group), and metallic character (decreases across a period, increases down a group). For example, atomic radius gets smaller from left to right in a period (e.g., Li > Be > B > C > N > O > F) and larger down a group (e.g., Li < Na < K < Rb < Cs).
2. Lighter vs. heavier elements
- Lighter elements (lower atomic mass) are found on the top-left of the periodic table. For example, hydrogen (H), helium (He), lithium (Li), and beryllium (Be) are light and in the top rows/left groups.
- Heavier elements (higher atomic mass) are found on the bottom-right (or lower rows in general). For example, radium (Ra), lead (Pb), and uranium (U) are heavy and in lower periods/groups.
3. Where is a period found?
A period is a horizontal row (left-to-right) on the periodic table. There are 7 periods (rows 1 to 7). For example, Period 1 contains H and He; Period 2 contains Li, Be, B, C, N, O, F, Ne.
4. Elements in the same period (horizontal row)
Elements in the same period have the same number of electron shells (energy levels). For example, all elements in Period 2 (Li to Ne) have 2 electron shells (n=2).
5. Finding the period number
The period number corresponds to the number of electron shells an element has. To find it, look at the element’s position: the row number (1–7) is the period number. For example, sodium (Na) is in Period 3 (row 3) and has 3 electron shells (n=1, n=2, n=3).
6. Where is a group found?
A group (or family) is a vertical column (top-to-bottom) on the periodic table. There are 18 groups (columns 1 to 18). For example, Group 1 (alkali metals: Li, Na, K, etc.) and Group 17 (halogens: F, Cl, Br, etc.) are vertical columns.
7. Elements in the same group (vertical column)
Elements in the same group have the same number of valence electrons (electrons in the outermost shell). For example, Group 1 (alkali metals: Li, Na, K) all have 1 valence electron; Group 17 (halogens: F, Cl, Br) all have 7 valence electrons.
8. What are valence electrons?
Valence electrons are the electrons in the outermost electron shell (highest energy level) of an atom. They determine an element’s chemical reactivity (e.g., bonding, ion formation). For example, oxygen (O) has 6 valence electrons (electrons in the n=2 shell).
These answers align with core periodic table trends in chemistry, focusing on electron structure, atomic properties, and spatial organization.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To answer these periodic table trend questions, we analyze each one based on chemistry concepts:
1. Properties of elements in a pattern
Elements show patterns in atomic radius (decreases across a period, increases down a group), ionization energy (increases across a period, decreases down a group), electronegativity (increases across a period, decreases down a group), and metallic character (decreases across a period, increases down a group). For example, atomic radius gets smaller from left to right in a period (e.g., Li > Be > B > C > N > O > F) and larger down a group (e.g., Li < Na < K < Rb < Cs).
2. Lighter vs. heavier elements
- Lighter elements (lower atomic mass) are found on the top-left of the periodic table. For example, hydrogen (H), helium (He), lithium (Li), and beryllium (Be) are light and in the top rows/left groups.
- Heavier elements (higher atomic mass) are found on the bottom-right (or lower rows in general). For example, radium (Ra), lead (Pb), and uranium (U) are heavy and in lower periods/groups.
3. Where is a period found?
A period is a horizontal row (left-to-right) on the periodic table. There are 7 periods (rows 1 to 7). For example, Period 1 contains H and He; Period 2 contains Li, Be, B, C, N, O, F, Ne.
4. Elements in the same period (horizontal row)
Elements in the same period have the same number of electron shells (energy levels). For example, all elements in Period 2 (Li to Ne) have 2 electron shells (n=2).
5. Finding the period number
The period number corresponds to the number of electron shells an element has. To find it, look at the element’s position: the row number (1–7) is the period number. For example, sodium (Na) is in Period 3 (row 3) and has 3 electron shells (n=1, n=2, n=3).
6. Where is a group found?
A group (or family) is a vertical column (top-to-bottom) on the periodic table. There are 18 groups (columns 1 to 18). For example, Group 1 (alkali metals: Li, Na, K, etc.) and Group 17 (halogens: F, Cl, Br, etc.) are vertical columns.
7. Elements in the same group (vertical column)
Elements in the same group have the same number of valence electrons (electrons in the outermost shell). For example, Group 1 (alkali metals: Li, Na, K) all have 1 valence electron; Group 17 (halogens: F, Cl, Br) all have 7 valence electrons.
8. What are valence electrons?
Valence electrons are the electrons in the outermost electron shell (highest energy level) of an atom. They determine an element’s chemical reactivity (e.g., bonding, ion formation). For example, oxygen (O) has 6 valence electrons (electrons in the n=2 shell).
These answers align with core periodic table trends in chemistry, focusing on electron structure, atomic properties, and spatial organization.