QUESTION IMAGE
Question
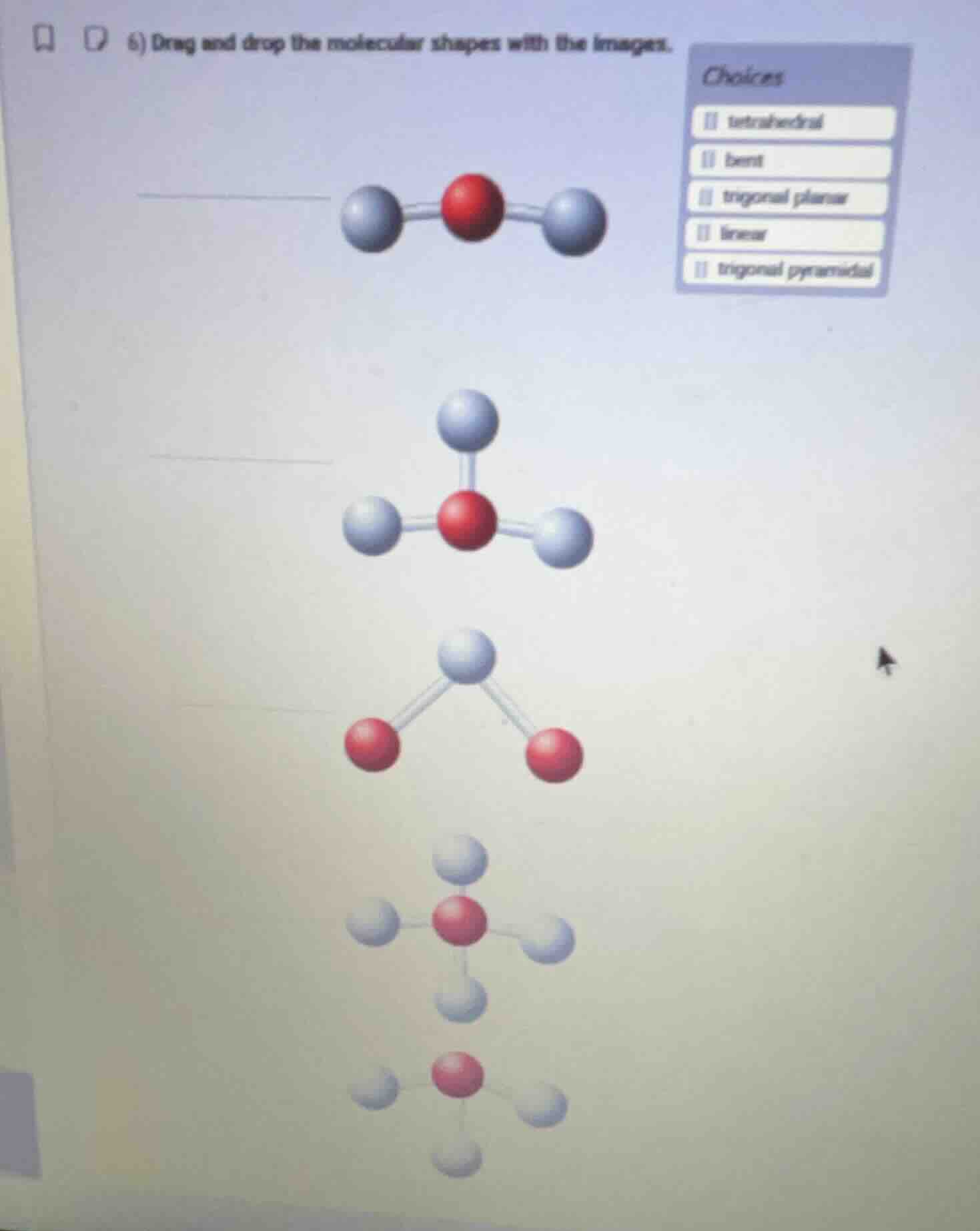
- drag and drop the molecular shapes with the images. choices: tetrahedral, bent, trigonal planar, linear, trigonal pyramidal
Brief Explanations
- The first molecule has a central atom bonded to two outer atoms in a straight line, matching linear shape.
- The second molecule has a central atom bonded to three outer atoms in a flat, triangular arrangement, matching trigonal planar shape.
- The third molecule has a central atom bonded to two outer atoms with a non-linear angle, matching bent shape.
- The fourth molecule has a central atom bonded to four outer atoms in a 3D triangular-based pyramid arrangement, matching tetrahedral shape.
- The fifth molecule has a central atom bonded to three outer atoms in a 3D pyramid shape, matching trigonal pyramidal shape.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Linear
- Trigonal planar
- Bent
- Tetrahedral
- Trigonal pyramidal