QUESTION IMAGE
Question
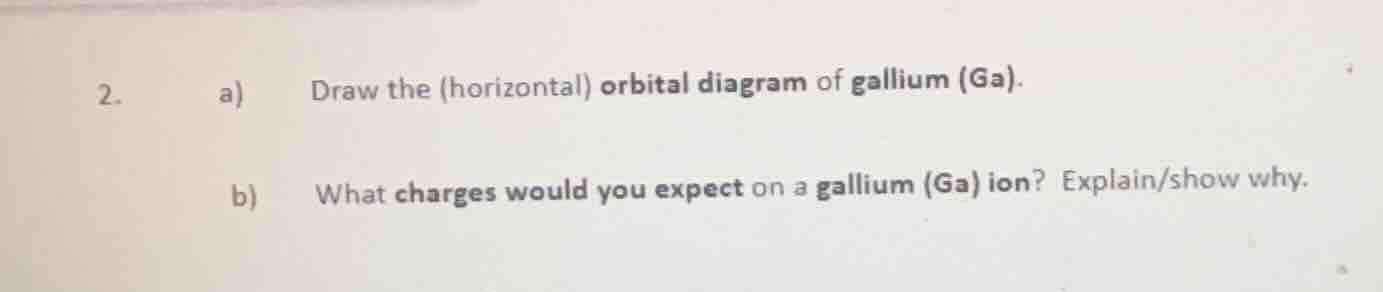
- a) draw the (horizontal) orbital diagram of gallium (ga). b) what charges would you expect on a gallium (ga) ion? explain/show why.
Part (a)
Step 1: Determine Electron Configuration
Gallium (Ga) has an atomic number of 31. The electron configuration is \(1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^{10} 4p^1\).
Step 2: Draw Orbital Diagram
- \(1s\): \(\uparrow\downarrow\)
- \(2s\): \(\uparrow\downarrow\)
- \(2p\): \(\uparrow\downarrow\ \uparrow\downarrow\ \uparrow\downarrow\)
- \(3s\): \(\uparrow\downarrow\)
- \(3p\): \(\uparrow\downarrow\ \uparrow\downarrow\ \uparrow\downarrow\)
- \(4s\): \(\uparrow\downarrow\)
- \(3d\): \(\uparrow\downarrow\ \uparrow\downarrow\ \uparrow\downarrow\ \uparrow\downarrow\ \uparrow\downarrow\)
- \(4p\): \(\uparrow\) (with two empty slots: \(\ \ \ \))
Part (b)
Gallium is in Group 13 of the periodic table. Elements in Group 13 typically have 3 valence electrons (from \(4s^2 4p^1\)). To achieve a stable electron configuration (like a noble gas), Ga can lose 3 electrons. When it loses 3 electrons, it forms a \(Ga^{3+}\) ion. This is because losing 3 electrons gives it the electron configuration of argon (a noble gas), which is stable. It can also potentially lose 1 electron (from the \(4p\) orbital) to have a filled \(3d\) and \(4s\) subshell, forming \(Ga^+\), but the most common and stable charge is \(3+\) due to the tendency to lose all 3 valence electrons to reach a noble gas configuration.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
(a):
The orbital diagram for Ga is:
\(1s\): \(\uparrow\downarrow\)
\(2s\): \(\uparrow\downarrow\)
\(2p\): \(\uparrow\downarrow\ \uparrow\downarrow\ \uparrow\downarrow\)
\(3s\): \(\uparrow\downarrow\)
\(3p\): \(\uparrow\downarrow\ \uparrow\downarrow\ \uparrow\downarrow\)
\(4s\): \(\uparrow\downarrow\)
\(3d\): \(\uparrow\downarrow\ \uparrow\downarrow\ \uparrow\downarrow\ \uparrow\downarrow\ \uparrow\downarrow\)
\(4p\): \(\uparrow\ \ \ \ \)