QUESTION IMAGE
Question
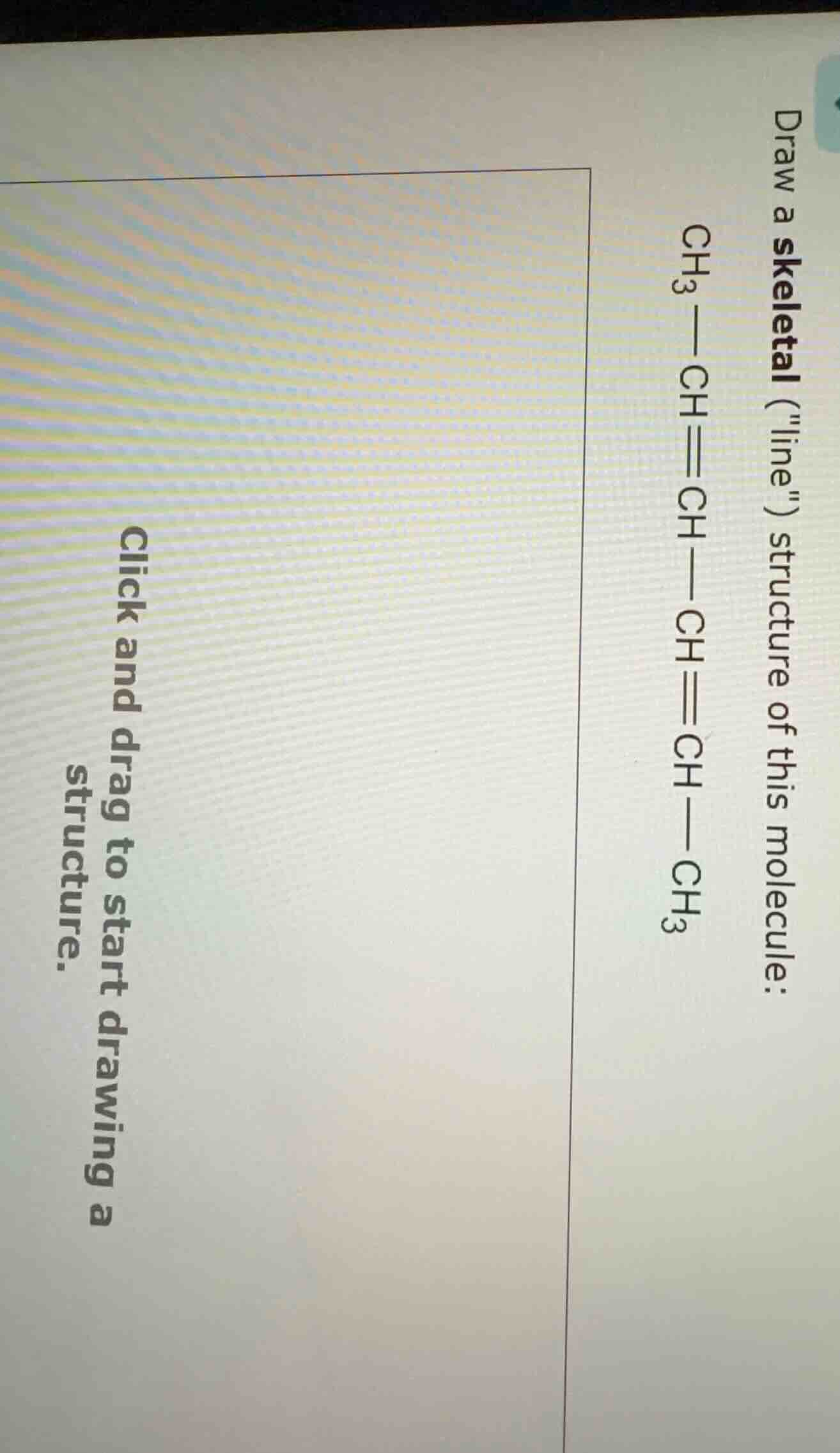
draw a skeletal (\line\) structure of this molecule:
ch₃—ch≡ch—ch≡ch—ch₃
click and drag to start drawing a structure.
Step1: Identify the carbon chain and bonds
The molecule is \( \text{CH}_3 - \text{CH} \equiv \text{CH} - \text{CH} \equiv \text{CH} - \text{CH}_3 \). In skeletal (line) structure, carbon atoms are represented by the vertices of the lines, and hydrogen atoms attached to carbons are omitted (except for terminal \( \text{CH}_3 \) which can be shown or implied, but in skeletal structure, terminal carbons with \( \text{CH}_3 \) are often shown as a line ending with a methyl group, but more simply, we represent the carbon - carbon bonds. The triple bonds and single bonds are maintained.
The carbon chain has 6 carbon atoms. The first carbon is \( \text{CH}_3 \), then a carbon with a triple bond to the next carbon, then a single bond to a carbon with a triple bond to the next carbon, and finally a \( \text{CH}_3 \).
Step2: Draw the skeletal structure
- Start with a line for the carbon - carbon bonds. The first carbon (from the left \( \text{CH}_3 \)) is a vertex, then a triple bond (represented by three parallel lines) to the next carbon, then a single bond (one line) to the next carbon, then a triple bond to the next carbon, then a single bond to the last carbon (which has \( \text{CH}_3 \)).
In skeletal structure, it can be drawn as: \( \text{CH}_3 - \ce{C\equiv C - C\equiv C - CH_3} \), but in line (skeletal) structure, we can represent it as:
CH3
|
C≡C - C≡C - CH3Wait, actually, the correct skeletal (line) structure for \( \text{CH}_3 - \text{CH} \equiv \text{CH} - \text{CH} \equiv \text{CH} - \text{CH}_3 \) is:
We can write it as a chain where the carbons are connected as follows: The first carbon (with \( \text{CH}_3 \)) is connected via a single bond to a carbon with a triple bond to the next carbon, then a single bond to a carbon with a triple bond to the next carbon, then a single bond to the last carbon (with \( \text{CH}_3 \)). In line structure, we represent the carbon - carbon bonds, so the structure is:
\( \text{CH}_3 - \ce{C\equiv C - C\equiv C - CH_3} \) can be drawn as:
CH3---C≡C---C≡C---CH3But in the standard skeletal (line) structure, we omit the \( \text{CH}_3 \) labels for non - terminal carbons (but here the terminal carbons have \( \text{CH}_3 \)). So the skeletal structure is a chain with two triple bonds and single bonds between the carbons with triple bonds, and methyl groups at the ends.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The skeletal (line) structure of the molecule \( \text{CH}_3 - \text{CH} \equiv \text{CH} - \text{CH} \equiv \text{CH} - \text{CH}_3 \) is \( \text{CH}_3 - \ce{C\equiv C - C\equiv C - CH_3} \) (or in line - angle notation: \( \text{CH}_3\ce{-C\equiv C - C\equiv C - CH_3} \) with the carbon - carbon bonds represented as lines, where the triple bonds are three parallel lines and single bonds are one line, and the \( \text{CH}_3 \) groups are at the ends). In a more simplified line (skeletal) drawing, it can be depicted as:
CH3---C≡C---C≡C---CH3(Note: If using a drawing tool, you would draw a chain with two triple bonds between the appropriate carbon atoms and methyl groups at the terminal carbons.)