QUESTION IMAGE
Question
electrons and the periodic table
quiz complete
1 2 3 4 5 6 7 8 9 10
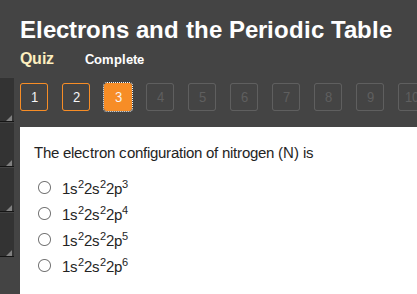
the electron configuration of nitrogen (n) is
○ 1s²2s²2p³
○ 1s²2s²2p⁴
○ 1s²2s²2p⁵
○ 1s²2s²2p⁶
by Aufbau principle
Fill 1s first (holds 2 electrons): $1s^2$ (2 electrons used, 5 left)
Fill 2s next (holds 2 electrons): $2s^2$ (4 electrons used, 3 left)
Fill 2p (holds 6 electrons, use remaining 3): $2p^3$
Step3: Combine orbitals
Combine the filled orbitals: $1s^22s^22p^3$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
by Aufbau principle
Fill 1s first (holds 2 electrons): $1s^2$ (2 electrons used, 5 left)
Fill 2s next (holds 2 electrons): $2s^2$ (4 electrons used, 3 left)
Fill 2p (holds 6 electrons, use remaining 3): $2p^3$
Step3: Combine orbitals
Combine the filled orbitals: $1s^22s^22p^3$