QUESTION IMAGE
Question
electrons and the periodic table
quiz complete
1 2 3 4 5 6 7 8 9 10
time remainin
57:36
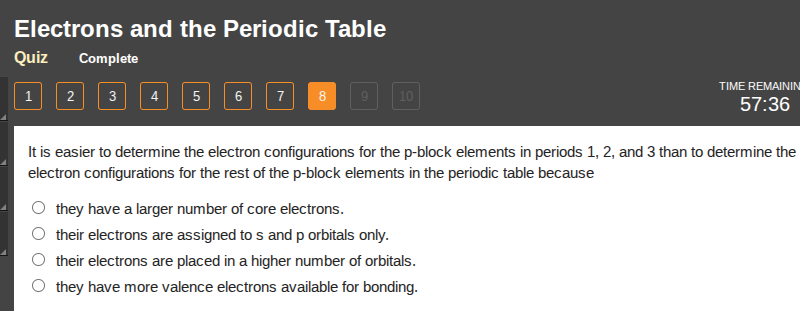
it is easier to determine the electron configurations for the p-block elements in periods 1, 2, and 3 than to determine the electron configurations for the rest of the p-block elements in the periodic table because
○ they have a larger number of core electrons.
○ their electrons are assigned to s and p orbitals only.
○ their electrons are placed in a higher number of orbitals.
○ they have more valence electrons available for bonding.
als to complicate configurations). Later p-block elements have filled inner d/f orbitals, making electron configuration determination more complex. The other options are incorrect: they have fewer core electrons, lower orbital counts, and valence electron count does not simplify configuration determination.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
als to complicate configurations). Later p-block elements have filled inner d/f orbitals, making electron configuration determination more complex. The other options are incorrect: they have fewer core electrons, lower orbital counts, and valence electron count does not simplify configuration determination.