QUESTION IMAGE
Question
experimental adjustment using the beer-lambert law
determination of percent copper
a quality assurance chemist runs an analysis to determine the
percent copper in a sample of brass used for plumbing fixtures.
she dissolves the sample using nitric acid and runs a
spectrophotometric analysis against a calibration of blue
copper(ii) nitrate solutions.
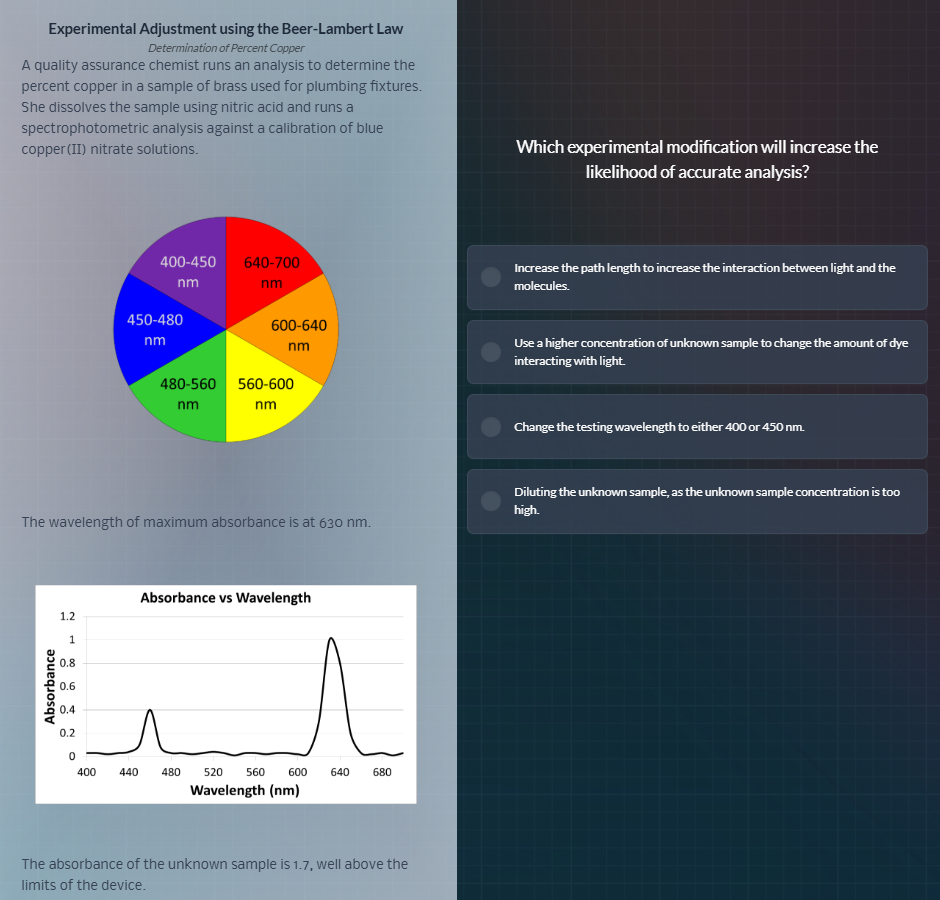
the wavelength of maximum absorbance is at 630 nm.
absorbance vs wavelength
the absorbance of the unknown sample is 1.7, well above the
limits of the device.
which experimental modification will increase the
likelihood of accurate analysis?
increase the path length to increase the interaction between light and the
molecules.
use a higher concentration of unknown sample to change the amount of dye
interacting with light.
change the testing wavelength to either 400 or 450 nm.
diluting the unknown sample, as the unknown sample concentration is too
high.
The Beer-Lambert Law relies on absorbance values within the device's linear range for accurate results. The unknown sample has an absorbance of 1.7, which is above the device's limits. Diluting the sample will lower its concentration, bringing the absorbance into the valid range for accurate analysis. The other options are incorrect: increasing path length would raise absorbance further, higher concentration would worsen the issue, and shifting wavelength away from the 630 nm maximum reduces sensitivity and accuracy.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Diluting the unknown sample, as the unknown sample concentration is too high.