QUESTION IMAGE
Question
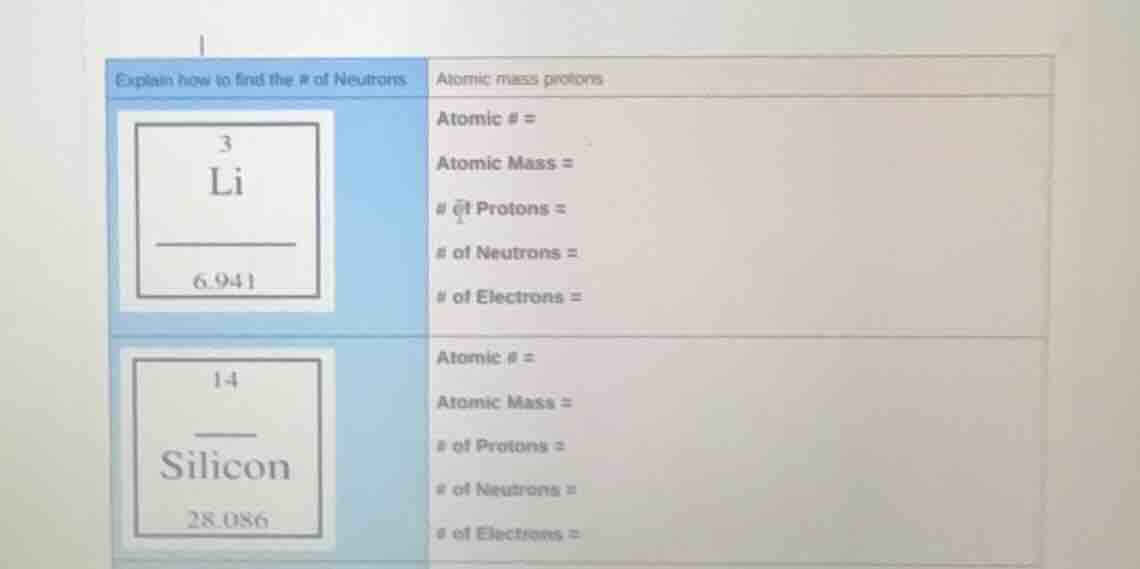
explain how to find the # of neutrons atomic mass protons
3
li
6.941
atomic # =
atomic mass =
of protons =
of neutrons =
of electrons =
14
silicon
28.086
atomic # =
atomic mass =
of protons =
of neutrons =
of electrons =
For Lithium (Li):
Step1: Recall Atomic Number
Atomic number (Z) is the number of protons, and for Li, the atomic number is 3 (from the top left, which is the atomic number). So Atomic # = 3.
Step2: Recall Atomic Mass
Atomic mass is the bottom number, so Atomic Mass = 6.941 (we can round it to 7 for neutron calculation as neutrons are calculated from mass number, which is the rounded atomic mass).
Step3: Number of Protons
In a neutral atom, number of protons = atomic number, so # of Protons = 3.
Step4: Number of Neutrons
Neutrons (n) = Mass number (A) - Atomic number (Z). Mass number A is the rounded atomic mass, so A = 7, Z = 3. Thus, n = 7 - 3 = 4. So # of Neutrons = 4.
Step5: Number of Electrons
In a neutral atom, number of electrons = number of protons, so # of Electrons = 3.
For Silicon (Si):
Step1: Recall Atomic Number
The atomic number of silicon is 14 (from the top left, atomic number). So Atomic # = 14.
Step2: Recall Atomic Mass
Atomic mass is 28.086 (bottom number), so Atomic Mass = 28.086 (we can take mass number A = 28 as it's the rounded atomic mass).
Step3: Number of Protons
Number of protons = atomic number, so # of Protons = 14.
Step4: Number of Neutrons
Neutrons (n) = A - Z. A = 28, Z = 14. So n = 28 - 14 = 14. Thus, # of Neutrons = 14.
Step5: Number of Electrons
In a neutral atom, electrons = protons, so # of Electrons = 14.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
(for each part):
Lithium:
Atomic # = 3
Atomic Mass = 6.941
of Protons = 3
of Neutrons = 4
of Electrons = 3
Silicon:
Atomic # = 14
Atomic Mass = 28.086