QUESTION IMAGE
Question
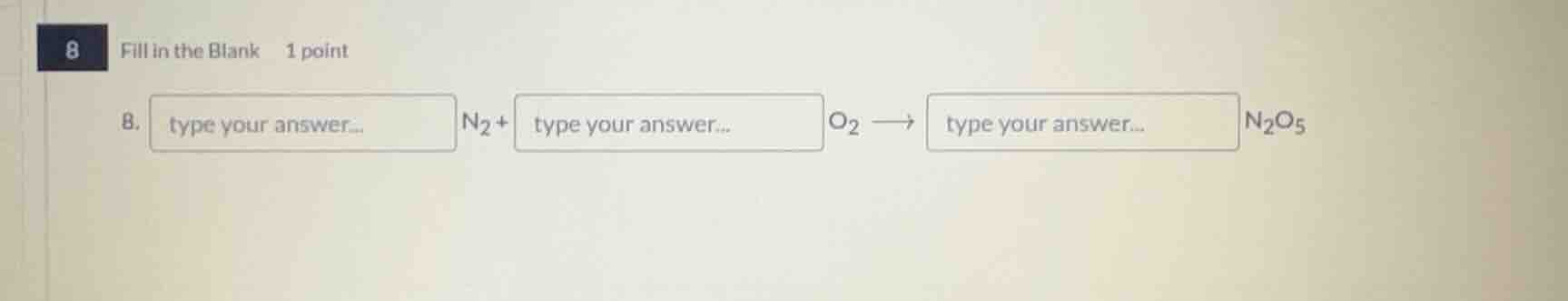
8 fill in the blank 1 point 8. type your answer... n₂ + type your answer... o₂ → type your answer... n₂o₅
Step1: Balance nitrogen atoms
On the left side, we have \( N_2 \), and on the right side, we have \( N_2O_5 \) (2 N atoms). So N is balanced for now.
Step2: Balance oxygen atoms
Right side: \( N_2O_5 \) has 5 O atoms. Left side: \( O_2 \) has 2 O atoms. Let the coefficient of \( O_2 \) be \( x \) and \( N_2O_5 \) be \( y \). For O: \( 2x = 5y \). Let's find the least common multiple of 2 and 5, which is 10. So \( x=\frac{5}{2} \), \( y = 2 \)? Wait, no, we want whole number coefficients. Multiply the entire equation by 2 to eliminate fractions.
Step3: Adjust coefficients for whole numbers
Multiply each term by 2: \( 2N_2 + 5O_2
ightarrow 2N_2O_5 \)? Wait, no, wait. Wait, original left: \( N_2 \) and \( O_2 \), right: \( N_2O_5 \). Let's balance step by step.
Let the equation be \( aN_2 + bO_2
ightarrow cN_2O_5 \).
For N: \( 2a = 2c \) (since left has 2a N, right has 2c N) ⇒ \( a = c \).
For O: \( 2b = 5c \). Let's take \( c = 2 \), then \( 2b = 10 \) ⇒ \( b = 5 \), and \( a = 2 \)? Wait, no, wait: if \( a = 1 \), then \( c = 1 \), so \( 2b = 5(1) \) ⇒ \( b = \frac{5}{2} \). But we need whole numbers, so multiply all coefficients by 2: \( 2N_2 + 5O_2
ightarrow 2N_2O_5 \)? Wait, no, that would give 4 N on left and 4 N on right (22=4), and 10 O on left (52=10) and 10 O on right (2*5=10). Wait, but the problem's right side is \( N_2O_5 \), not \( 2N_2O_5 \). Wait, maybe I made a mistake. Wait, let's check again.
Wait, the product is \( N_2O_5 \) (1 molecule). So for N: left has \( N_2 \) (2 N), right has \( N_2O_5 \) (2 N). So N is balanced with coefficient 1 for \( N_2 \) and 1 for \( N_2O_5 \). Now O: right has 5 O, left has \( O_2 \) (2 O per molecule). So coefficient of \( O_2 \) should be \( \frac{5}{2} \). But we can't have fractions, so multiply the entire equation by 2 to get whole numbers: \( 2N_2 + 5O_2
ightarrow 2N_2O_5 \). Wait, but the problem's blanks are for coefficients. Let's see the original blanks: first blank (coefficient of \( N_2 \)), second (coefficient of \( O_2 \)), third (coefficient of \( N_2O_5 \)).
Wait, let's do it properly. Let the equation be:
\( \text{[coefficient]} N_2 + \text{[coefficient]} O_2
ightarrow \text{[coefficient]} N_2O_5 \)
Balance N: left has 2[N2 coefficient], right has 2[N2O5 coefficient]. So [N2 coefficient] = [N2O5 coefficient].
Balance O: left has 2[O2 coefficient], right has 5[N2O5 coefficient]. So 2[O2] = 5[N2O5]. Let [N2O5] = 2 (to make O even), then [O2] = 5, [N2] = 2. Wait, but if [N2O5] = 1, then [O2] = 5/2, [N2] = 1. But we need whole numbers, so the balanced equation with whole numbers is \( 2N_2 + 5O_2
ightarrow 2N_2O_5 \)? No, wait, no: wait, \( N_2 + \frac{5}{2}O_2
ightarrow N_2O_5 \). Multiply all by 2: \( 2N_2 + 5O_2
ightarrow 2N_2O_5 \). But the problem's product is \( N_2O_5 \), not \( 2N_2O_5 \). Wait, maybe the problem allows fractional coefficients? No, usually we use whole numbers. Wait, maybe I messed up. Wait, let's check the number of atoms:
Left: \( N_2 \) (2 N) + \( O_2 \) (2 O per molecule). Right: \( N_2O_5 \) (2 N, 5 O).
So for O: 2[O2] = 5[N2O5]. Let [N2O5] = 2, then [O2] = 5, [N2] = 2. Then left: 22=4 N, 52=10 O. Right: 22=4 N, 25=10 O. That works. So the coefficients are 2, 5, 2? Wait, no, wait: \( 2N_2 + 5O_2
ightarrow 2N_2O_5 \). But the problem's right side is \( N_2O_5 \), not \( 2N_2O_5 \). Wait, maybe the problem has a typo, or maybe I'm overcomplicating. Wait, let's do it with the product as \( N_2O_5 \) (1 molecule). Then:
N: 2 (left) = 2 (right) → coefficient of \( N_2 \) is 1.
O: 2[O2] = 51 → [O2] = 5/2.
So th…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
First blank: 2, Second blank: 5, Third blank: 2