QUESTION IMAGE
Question
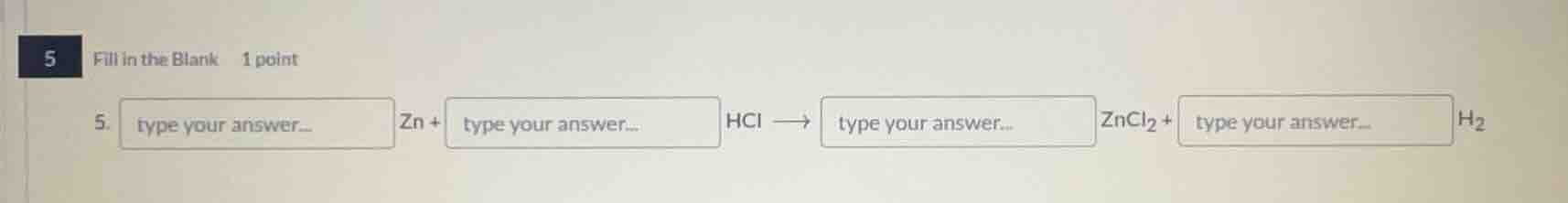
5 fill in the blank 1 point 5. type your answer... zn + type your answer... hcl → type your answer... zncl₂ + type your answer... h₂
Step1: Identify the reaction type
This is a single - displacement reaction where zinc (Zn) reacts with hydrochloric acid (HCl). The general form of a single - displacement reaction is \(A + BC
ightarrow AC + B\), where in this case \(A = Zn\), \(B = H\) and \(C=Cl\).
Step2: Balance the chemical equation
- Let's start with the unbalanced equation: \(Zn+HCl
ightarrow ZnCl_{2}+H_{2}\)
- First, balance the chlorine atoms. On the right side, we have 2 chlorine atoms in \(ZnCl_{2}\), so we need 2 moles of \(HCl\) on the left side. So now the equation becomes \(Zn + 2HCl
ightarrow ZnCl_{2}+H_{2}\)
- Now check the zinc and hydrogen atoms. The number of zinc atoms on the left (1) and right (1) is balanced. The number of hydrogen atoms on the left (2 in \(2HCl\)) and on the right (2 in \(H_{2}\)) is balanced. Also, there is no coefficient needed for \(Zn\) as it is already balanced. So the balanced equation is \(1Zn + 2HCl
ightarrow 1ZnCl_{2}+1H_{2}\)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The coefficients (from left to right) are 1, 2, 1, 1. So the blanks should be filled with 1, 2, 1, 1 respectively.