QUESTION IMAGE
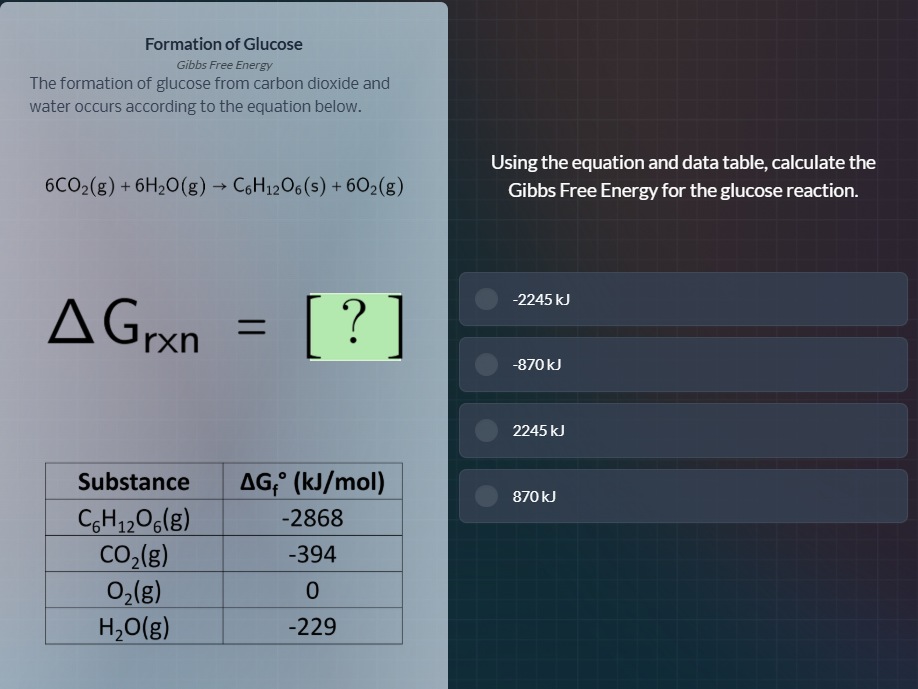
Question
formation of glucose
gibbs free energy
the formation of glucose from carbon dioxide and
water occurs according to the equation below.
6co₂(g) + 6h₂o(g) → c₆h₁₂o₆(s) + 6o₂(g)
δgᵣₓₙ =?
substance | δgᵣ° (kj/mol)
c₆h₁₂o₆(g) | -2868
co₂(g) | -394
o₂(g) | 0
h₂o(g) | -229
using the equation and data table, calculate the
gibbs free energy for the glucose reaction.
-2245 kj
-870 kj
2245 kj
870 kj
Step1: Recall the formula for Gibbs Free Energy of reaction
The formula for the Gibbs Free Energy change of a reaction ($\Delta G_{rxn}$) is given by the sum of the Gibbs Free Energy of formation ($\Delta G_f^{\circ}$) of the products minus the sum of the Gibbs Free Energy of formation of the reactants, multiplied by their respective stoichiometric coefficients. Mathematically, it is:
$$\Delta G_{rxn} = \sum (n \times \Delta G_f^{\circ}_{\text{products}}) - \sum (m \times \Delta G_f^{\circ}_{\text{reactants}})$$
where $n$ and $m$ are the stoichiometric coefficients of the products and reactants respectively.
Step2: Identify the products and reactants with their stoichiometric coefficients
For the reaction: $6\text{CO}_2(g) + 6\text{H}_2\text{O}(g)
ightarrow \text{C}_6\text{H}_{12}\text{O}_6(s) + 6\text{O}_2(g)$
- Products: $\text{C}_6\text{H}_{12}\text{O}_6(s)$ (stoichiometric coefficient $n = 1$) and $\text{O}_2(g)$ (stoichiometric coefficient $n = 6$)
- Reactants: $\text{CO}_2(g)$ (stoichiometric coefficient $m = 6$) and $\text{H}_2\text{O}(g)$ (stoichiometric coefficient $m = 6$)
Step3: Find the $\Delta G_f^{\circ}$ values for each substance
From the data table:
- $\Delta G_f^{\circ}(\text{C}_6\text{H}_{12}\text{O}_6(s)) = -2868$ kJ/mol (Wait, the table has $\text{C}_6\text{H}_{12}\text{O}_6(g)$ but the product is solid? Wait, maybe it's a typo, but let's check the table again. Wait the table says $\text{C}_6\text{H}_{12}\text{O}_6(g)$ with $\Delta G_f^{\circ} = -2868$ kJ/mol. Maybe it's a typo and should be solid, but we'll use the given values.
- $\Delta G_f^{\circ}(\text{O}_2(g)) = 0$ kJ/mol
- $\Delta G_f^{\circ}(\text{CO}_2(g)) = -394$ kJ/mol
- $\Delta G_f^{\circ}(\text{H}_2\text{O}(g)) = -229$ kJ/mol
Step4: Calculate the sum of $\Delta G_f^{\circ}$ for products
For products:
- $\text{C}_6\text{H}_{12}\text{O}_6(s)$ (or gas as per table): $n = 1$, so $1 \times (-2868)$ kJ/mol
- $\text{O}_2(g)$: $n = 6$, so $6 \times 0$ kJ/mol
Sum of products: $1\times(-2868) + 6\times0 = -2868$ kJ
Step5: Calculate the sum of $\Delta G_f^{\circ}$ for reactants
For reactants:
- $\text{CO}_2(g)$: $m = 6$, so $6 \times (-394)$ kJ/mol
- $\text{H}_2\text{O}(g)$: $m = 6$, so $6 \times (-229)$ kJ/mol
First, calculate for $\text{CO}_2$: $6\times(-394) = -2364$ kJ
For $\text{H}_2\text{O}$: $6\times(-229) = -1374$ kJ
Sum of reactants: $-2364 + (-1374) = -3738$ kJ
Step6: Calculate $\Delta G_{rxn}$
Using the formula:
$$\Delta G_{rxn} = (\text{Sum of products}) - (\text{Sum of reactants})$$
Substitute the values:
$$\Delta G_{rxn} = (-2868) - (-3738)$$
$$\Delta G_{rxn} = -2868 + 3738$$
$$\Delta G_{rxn} = 870$$ Wait, that can't be right. Wait, maybe I mixed up products and reactants. Wait the formula is $\sum$ (products) - $\sum$ (reactants). Wait let's re-express:
Wait the reaction is $6\text{CO}_2 + 6\text{H}_2\text{O}
ightarrow \text{C}_6\text{H}_{12}\text{O}_6 + 6\text{O}_2$
So products are $\text{C}_6\text{H}_{12}\text{O}_6$ (1 mol) and $\text{O}_2$ (6 mol)
Reactants are $\text{CO}_2$ (6 mol) and $\text{H}_2\text{O}$ (6 mol)
So $\sum \Delta G_f^{\circ}_{\text{products}} = [\Delta G_f^{\circ}(\text{C}_6\text{H}_{12}\text{O}_6) + 6\times\Delta G_f^{\circ}(\text{O}_2)]$
$\sum \Delta G_f^{\circ}_{\text{reactants}} = [6\times\Delta G_f^{\circ}(\text{CO}_2) + 6\times\Delta G_f^{\circ}(\text{H}_2\text{O})]$
Then $\Delta G_{rxn} = \sum \text{products} - \sum \text{reactants}$
Plugging in the values:
$\Delta G_f^{\circ}(\text{C}_6\text{H}_{12}\text{O}_6) = -2868$ (from table, even though it's gas, maybe the problem has a typo)…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
870 kJ (corresponding to the option "870 kJ")