QUESTION IMAGE
Question
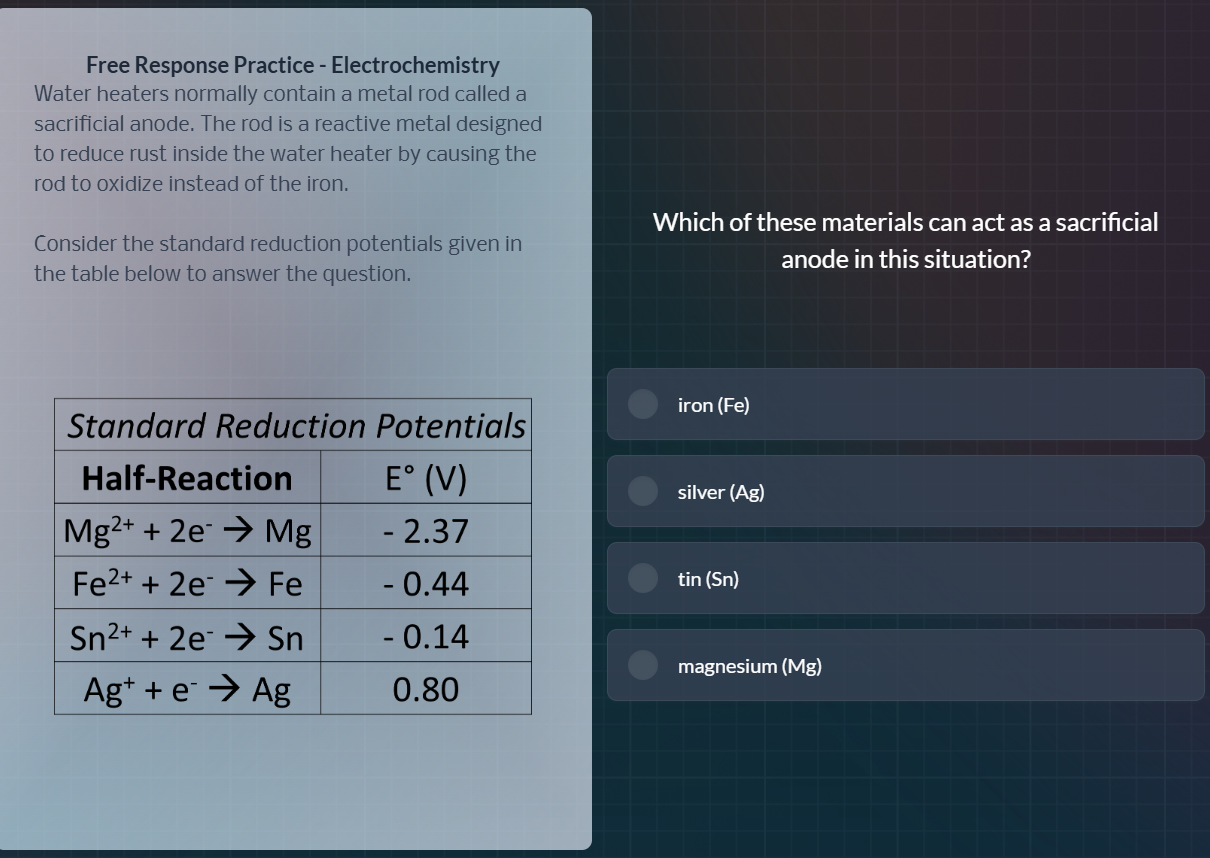
free response practice - electrochemistry
water heaters normally contain a metal rod called a
sacrificial anode. the rod is a reactive metal designed
to reduce rust inside the water heater by causing the
rod to oxidize instead of the iron.
consider the standard reduction potentials given in
the table below to answer the question.
standard reduction potentials
half - reaction | e° (v)
mg²⁺ + 2e⁻ → mg | - 2.37
fe²⁺ + 2e⁻ → fe | - 0.44
sn²⁺ + 2e⁻ → sn | - 0.14
ag⁺ + e⁻ → ag | 0.80
which of these materials can act as a sacrificial
anode in this situation?
iron (fe)
silver (ag)
tin (sn)
magnesium (mg)
A sacrificial anode must be a metal more reactive (more easily oxidized) than iron. Oxidation is the reverse of reduction, so a metal with a more negative standard reduction potential (E°) than Fe will be more easily oxidized. The E° for Fe²⁺ + 2e⁻ → Fe is -0.44 V. Comparing the given metals:
- Fe: Same as the metal we want to protect, can’t sacrifice itself.
- Ag: E° = 0.80 V (more positive than -0.44), less reactive.
- Sn: E° = -0.14 V (less negative than -0.44), less reactive.
- Mg: E° = -2.37 V (more negative than -0.44), more reactive.
Thus, Mg is more easily oxidized than Fe, so it can act as the sacrificial anode.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
magnesium (Mg)