QUESTION IMAGE
Question
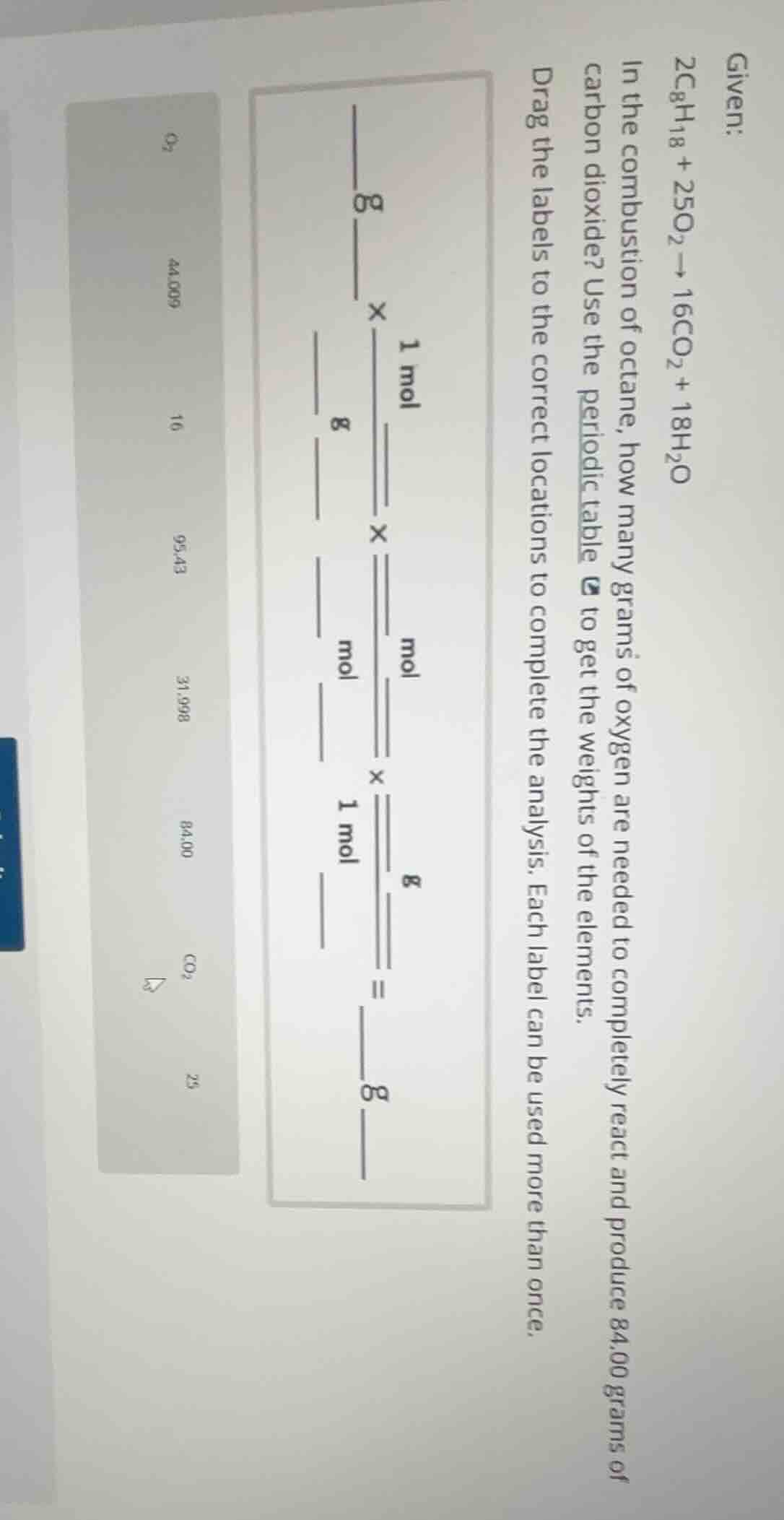
given:
$2\ce{c8h18} + 25\ce{o2} \
ightarrow 16\ce{co2} + 18\ce{h2o}$
in the combustion of octane, how many grams of oxygen are needed to completely react and produce 84.00 grams of carbon dioxide? use the periodic table to get the weights of the elements.
drag the labels to the correct locations to complete the analysis. each label can be used more than once.
(there is an image of a calculation setup with blanks to fill and a list of labels on the left: $\ce{o2}$, 44.009, 16, 95.43, 31.998, 84.00, $\ce{co2}$, 25)
Step1: Start with given CO₂ mass
$84.00\ \text{g}\ \text{CO}_2$
Step2: Convert CO₂ to moles
$\frac{1\ \text{mol}\ \text{CO}_2}{44.009\ \text{g}\ \text{CO}_2}$
Step3: Mole ratio of O₂ to CO₂
$\frac{25\ \text{mol}\ \text{O}_2}{16\ \text{mol}\ \text{CO}_2}$
Step4: Convert O₂ moles to grams
$\frac{31.998\ \text{g}\ \text{O}_2}{1\ \text{mol}\ \text{O}_2}$
Step5: Calculate the final value
$84.00 \times \frac{1}{44.009} \times \frac{25}{16} \times 31.998$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$84.00\ \text{g}\ \text{CO}_2 \times \frac{1\ \text{mol}\ \text{CO}_2}{44.009\ \text{g}\ \text{CO}_2} \times \frac{25\ \text{mol}\ \text{O}_2}{16\ \text{mol}\ \text{CO}_2} \times \frac{31.998\ \text{g}\ \text{O}_2}{1\ \text{mol}\ \text{O}_2} = 95.43\ \text{g}\ \text{O}_2$
Filled sequence (left to right, top to bottom):
84.00, 44.009, 25, 16, 31.998, 95.43