QUESTION IMAGE
Question
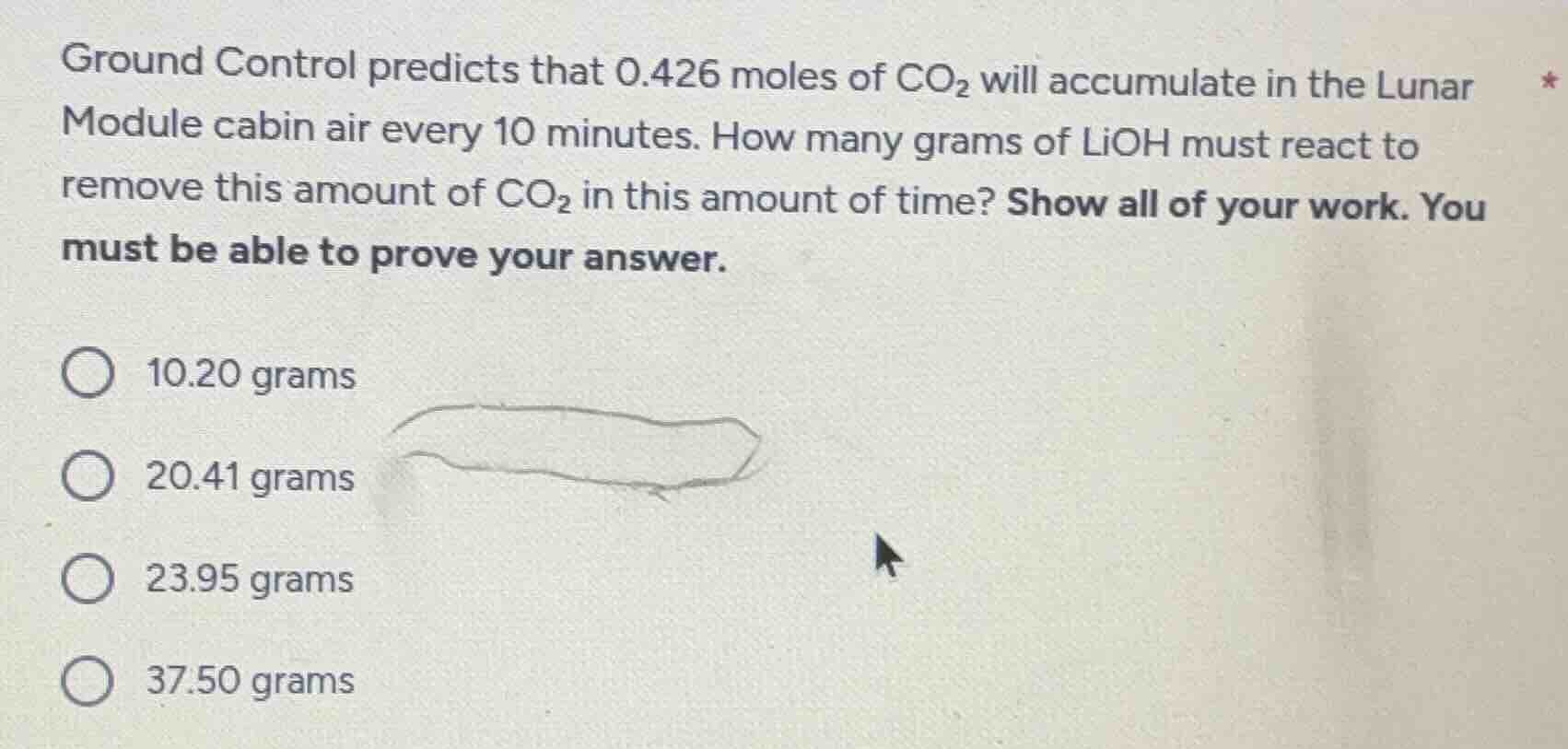
ground control predicts that 0.426 moles of co₂ will accumulate in the lunar module cabin air every 10 minutes. how many grams of lioh must react to remove this amount of co₂ in this amount of time? show all of your work. you must be able to prove your answer. 10.20 grams 20.41 grams 23.95 grams 37.50 grams
Step1: Write the balanced reaction
The reaction between \( \ce{LiOH} \) and \( \ce{CO_2} \) is \( 2\ce{LiOH} + \ce{CO_2}
ightarrow \ce{Li_2CO_3} + \ce{H_2O} \). From the reaction, 1 mole of \( \ce{CO_2} \) reacts with 2 moles of \( \ce{LiOH} \).
Step2: Calculate moles of \( \ce{LiOH} \)
Moles of \( \ce{CO_2} = 0.426 \) mol. Moles of \( \ce{LiOH} = 2\times \) moles of \( \ce{CO_2} = 2\times0.426 = 0.852 \) mol.
Step3: Calculate molar mass of \( \ce{LiOH} \)
Molar mass of \( \ce{Li} = 6.94 \) g/mol, \( \ce{O} = 16.00 \) g/mol, \( \ce{H} = 1.01 \) g/mol. Molar mass of \( \ce{LiOH} = 6.94 + 16.00 + 1.01 = 23.95 \) g/mol.
Step4: Calculate mass of \( \ce{LiOH} \)
Mass = moles \( \times \) molar mass = \( 0.852 \) mol \( \times23.95 \) g/mol \( \approx 20.41 \) g.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
20.41 grams (Option: 20.41 grams)