QUESTION IMAGE
Question
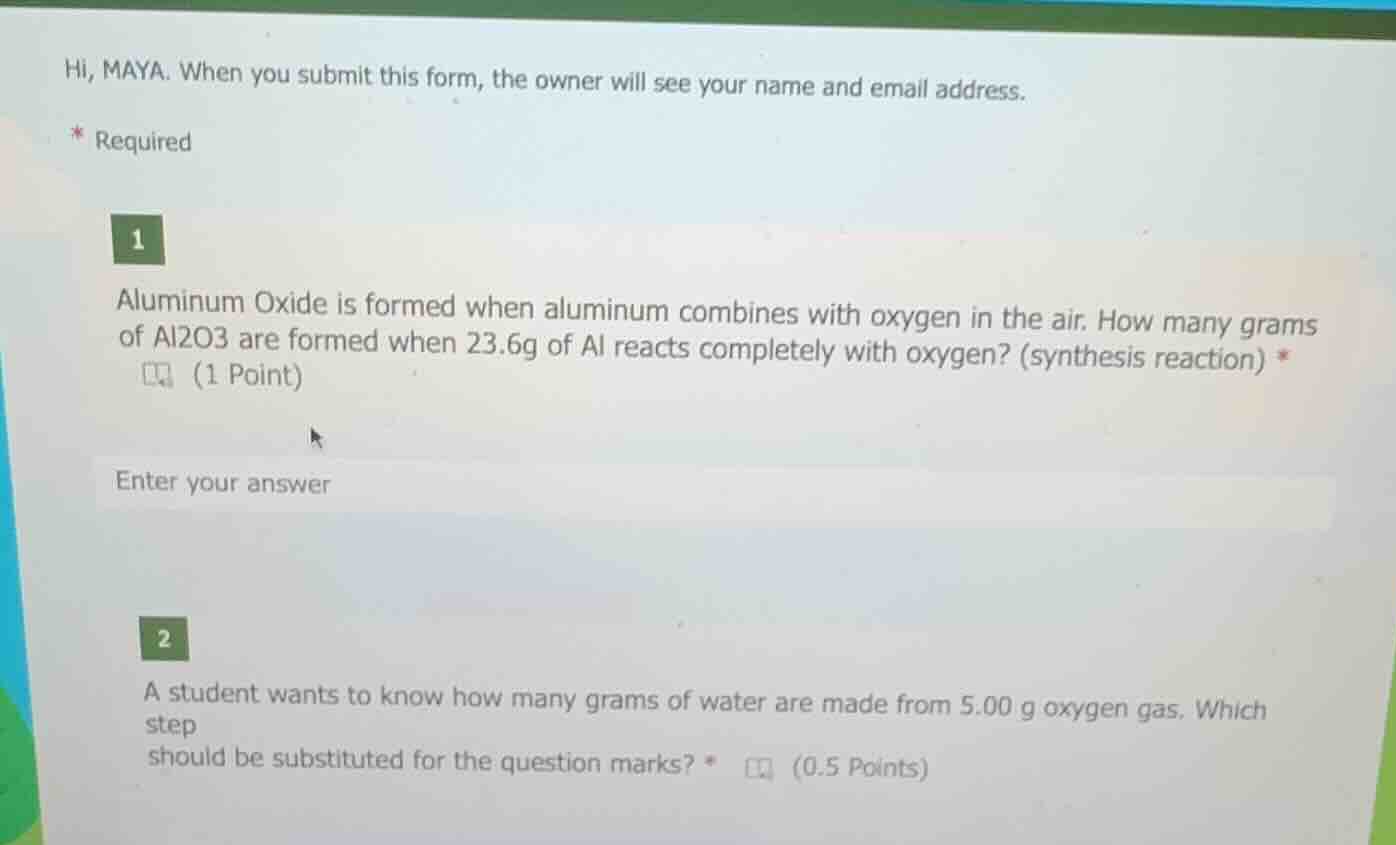
hi, maya. when you submit this form, the owner will see your name and email address.
- required
1
aluminum oxide is formed when aluminum combines with oxygen in the air. how many grams of al2o3 are formed when 23.6g of al reacts completely with oxygen? (synthesis reaction) *
(1 point)
enter your answer
2
a student wants to know how many grams of water are made from 5.00 g oxygen gas. which step
should be substituted for the question marks? *
(0.5 points)
Question 1
Step 1: Write the balanced chemical equation
The reaction between aluminum (Al) and oxygen (O₂) to form aluminum oxide (Al₂O₃) is:
Step 2: Calculate moles of Al
Molar mass of Al = \(26.98\ \text{g/mol}\).
Moles of Al = \(\frac{\text{mass of Al}}{\text{molar mass of Al}} = \frac{23.6\ \text{g}}{26.98\ \text{g/mol}} \approx 0.8747\ \text{mol}\).
Step 3: Relate moles of Al to moles of Al₂O₃
From the balanced equation, \(4\ \text{mol Al}
ightarrow 2\ \text{mol Al}_2\text{O}_3\) (or \(2\ \text{mol Al}
ightarrow 1\ \text{mol Al}_2\text{O}_3\)).
Moles of Al₂O₃ = \(\frac{0.8747\ \text{mol Al}}{2} \approx 0.43735\ \text{mol}\).
Step 4: Calculate molar mass of Al₂O₃
Molar mass of Al₂O₃ = \(2(26.98) + 3(16.00) = 53.96 + 48.00 = 101.96\ \text{g/mol}\).
Step 5: Calculate mass of Al₂O₃
Mass of Al₂O₃ = moles × molar mass = \(0.43735\ \text{mol} × 101.96\ \text{g/mol} \approx 44.6\ \text{g}\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(\approx 44.6\ \text{g}\) (or more precisely, ~44.5 - 44.7 g depending on rounding)
Question 2 (Incomplete, but assuming the typical stoichiometry problem for \( \text{O}_2
ightarrow \text{H}_2\text{O} \))
The reaction forming water is \(2\text{H}_2 + \text{O}_2
ightarrow 2\text{H}_2\text{O}\). For \(5.00\ \text{g O}_2\):
- Moles of \(\text{O}_2\): \(\frac{5.00\ \text{g}}{32.00\ \text{g/mol}}\) (molar mass of \(\text{O}_2 = 32.00\ \text{g/mol}\)).
- Mole ratio: \(1\ \text{mol O}_2
ightarrow 2\ \text{mol H}_2\text{O}\) (from balanced equation).
- Molar mass of \(\text{H}_2\text{O} = 18.02\ \text{g/mol}\), so mass of \(\text{H}_2\text{O}\) = moles of \(\text{H}_2\text{O} × 18.02\ \text{g/mol}\).
If the “question marks” refer to these steps (e.g., mole ratio, molar mass), the substitution would follow stoichiometry:
- Moles of \(\text{O}_2\): \(\frac{5.00}{32.00}\)
- Mole ratio: \(2\ \text{mol H}_2\text{O} / 1\ \text{mol O}_2\)
- Molar mass of \(\text{H}_2\text{O}\): \(18.02\ \text{g/mol}\)
(Note: The exact substitution depends on the specific format of the question’s “question marks,” but this outlines the stoichiometric steps.)