QUESTION IMAGE
Question
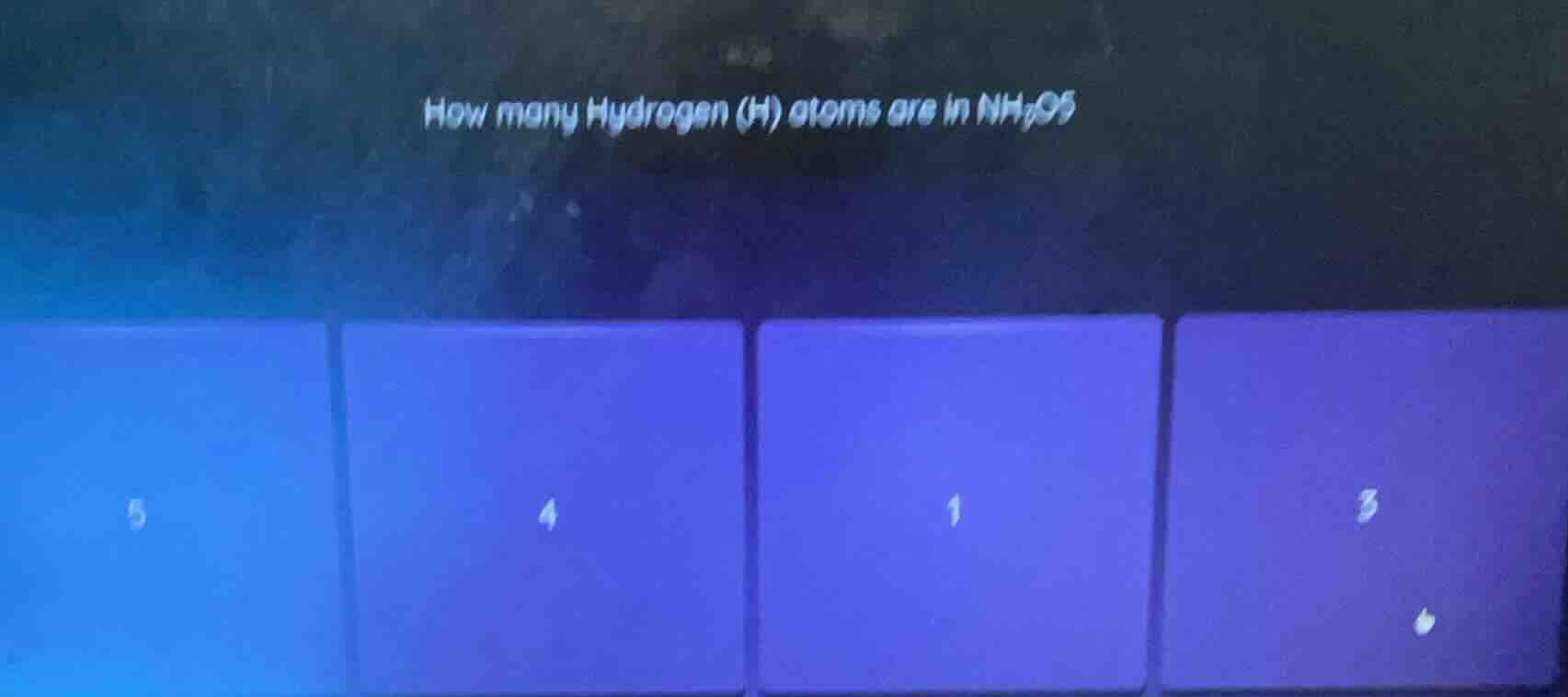
how many hydrogen (h) atoms are in nh₄os
Step1: Analyze the chemical formula
In the chemical formula \(NH_4OS\) (assuming it's a typo and should be \(NH_4OS\) or similar, but focusing on \(H\) subscript), the subscript of \(H\) is 4? Wait, no, wait, the formula is \(NH_4OS\)? Wait, no, the user wrote \(NH_4OS\)? Wait, no, the original is \(NH_4OS\)? Wait, no, looking at the question: "How many Hydrogen (H) atoms are in \(NH_4OS\)"? Wait, no, maybe it's \(NH_4O\) or \(NH_4S\)? Wait, no, the subscript for \(H\) is 4? Wait, no, wait, the formula is \(NH_4\) part? Wait, no, let's check: in a chemical formula, the subscript after an element tells the number of atoms. So in \(NH_4\) (like in \(NH_4^+\) or compounds with \(NH_4\) group), the number of \(H\) atoms is 4? Wait, no, wait the options have 4 as one of them. Wait, maybe the formula is \(NH_4OS\), so the \(H\) has a subscript of 4. Wait, let's re-express:
In a chemical formula, the number of atoms of an element is given by the subscript. For \(H\) in \(NH_4\) (the ammonium group), the subscript is 4. So the number of \(H\) atoms is 4.
Step2: Match with options
Looking at the options, one of them is 4. So the correct answer is the option with 4.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The option with "4" (assuming the options are labeled, e.g., if the second purple box is labeled B, then B. 4)