QUESTION IMAGE
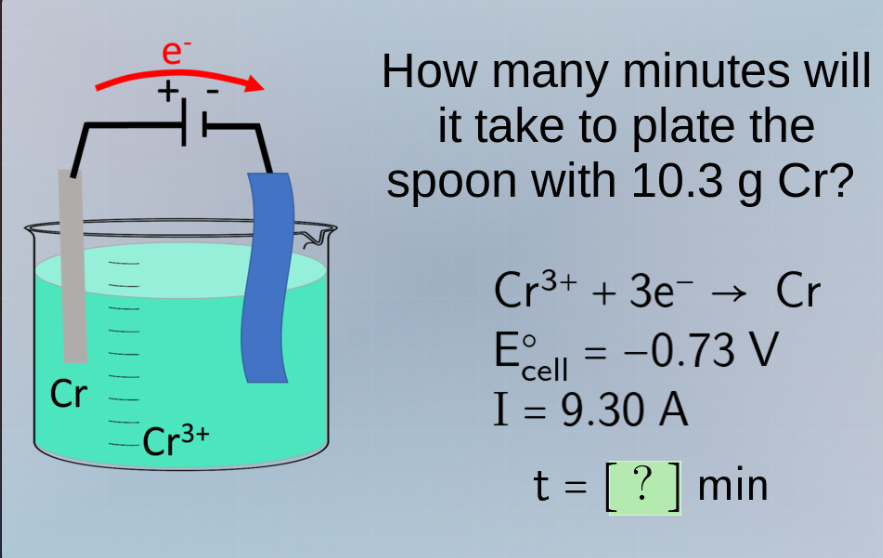
Question
how many minutes will it take to plate the spoon with 10.3 g cr?
cr³⁺ + 3e⁻ → cr
e°_cell = -0.73 v
i = 9.30 a
t = ? min
Step1: Calculate moles of Cr
Molar mass of Cr is 52.00 g/mol. Moles of Cr, \( n = \frac{mass}{molar\ mass} = \frac{10.3\ g}{52.00\ g/mol} \approx 0.1981\ mol \).
Step2: Determine moles of electrons
From the reaction \( \text{Cr}^{3+} + 3e^-
ightarrow \text{Cr} \), 3 moles of \( e^- \) are needed per mole of Cr. So moles of \( e^- \), \( n_{e^-} = 3 \times 0.1981\ mol = 0.5943\ mol \).
Step3: Calculate charge (in coulombs)
Using Faraday's constant (\( F = 96485\ C/mol\ e^- \)), charge \( Q = n_{e^-} \times F = 0.5943\ mol \times 96485\ C/mol \approx 57340\ C \).
Step4: Relate charge, current, and time
Current \( I = \frac{Q}{t} \), so \( t = \frac{Q}{I} \). Substituting values: \( t = \frac{57340\ C}{9.30\ A} \approx 6165.6\ s \).
Step5: Convert seconds to minutes
\( t (\text{min}) = \frac{6165.6\ s}{60\ s/min} \approx 102.76\ min \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( \approx 103 \) (or more precisely, around 102.8) minutes.