QUESTION IMAGE
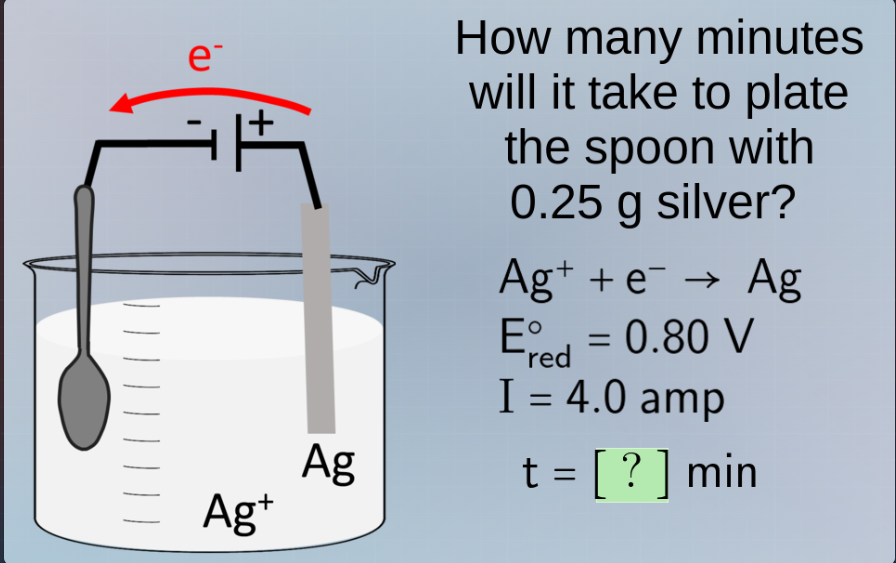
Question
how many minutes will it take to plate the spoon with 0.25 g silver? $\ce{ag+ + e- -> ag}$ $e_{\text{red}}^\circ = 0.80$ v $i = 4.0$ amp $t = ?$ min
Step1: Calculate moles of Ag
Molar mass of Ag is 107.87 g/mol. Moles of Ag, \( n = \frac{\text{mass}}{\text{molar mass}} = \frac{0.25\ \text{g}}{107.87\ \text{g/mol}} \approx 0.002317\ \text{mol} \).
Step2: Determine moles of electrons
From the reaction \( \text{Ag}^+ + \text{e}^-
ightarrow \text{Ag} \), 1 mole of Ag is formed from 1 mole of electrons. So moles of electrons, \( n_e = 0.002317\ \text{mol} \).
Step3: Calculate charge (Q)
Using Faraday's law, \( Q = n_e \times F \), where \( F = 96485\ \text{C/mol} \). So \( Q = 0.002317\ \text{mol} \times 96485\ \text{C/mol} \approx 223.6\ \text{C} \).
Step4: Relate charge to current and time
Current \( I = \frac{Q}{t} \), so \( t = \frac{Q}{I} \). Given \( I = 4.0\ \text{A} \), \( t = \frac{223.6\ \text{C}}{4.0\ \text{A}} = 55.9\ \text{s} \). Convert to minutes: \( t = \frac{55.9\ \text{s}}{60\ \text{s/min}} \approx 0.93\ \text{min} \)? Wait, no, wait, miscalculation. Wait, let's redo step 1: 0.25 g Ag, molar mass 107.87, so moles Ag = 0.25 / 107.87 ≈ 0.002317 mol. Moles of e⁻ is same as moles of Ag, so Q = n_e F = 0.002317 mol 96485 C/mol ≈ 223.6 C. Then t (in seconds) = Q / I = 223.6 C / 4.0 A = 55.9 s. Wait, that can't be right. Wait, no, wait, maybe I messed up the molar mass? Wait, no, 107.87 is correct. Wait, wait, 0.25 g is a small amount, but 4 A is a large current. Wait, no, wait, let's check again. Wait, 0.25 g Ag: moles = 0.25 / 107.87 ≈ 0.002317 mol. Each mole of Ag requires 1 mole of e⁻, so Q = 0.002317 mol 96485 C/mol ≈ 223.6 C. Time in seconds: 223.6 C / 4 A = 55.9 s, which is about 0.93 minutes. But that seems too fast. Wait, maybe I made a mistake in the reaction. Wait, the reaction is Ag⁺ + e⁻ → Ag, so 1 mole e⁻ per mole Ag. Wait, maybe the current is 4.0 amp, which is 4 C/s. So Q = It, so t = Q/I. Q is in coulombs. So let's recalculate moles of Ag: 0.25 g / 107.87 g/mol = 0.002317 mol. Moles of e⁻ = 0.002317 mol. Q = 0.002317 mol 96485 C/mol = 223.6 C. t = 223.6 C / 4 A = 55.9 s. Convert to minutes: 55.9 / 60 ≈ 0.93 min. Wait, but that seems too short. Wait, maybe the mass is 0.25 grams, but 4 A is 4 coulombs per second. Let's check with another approach. The formula for electroplating is: mass = (I t M) / (n F), where M is molar mass, n is moles of e⁻ per mole of product, F is Faraday constant. So rearranging for t: t = (mass n F) / (I M). Here, mass = 0.25 g, n = 1, F = 96485 C/mol, I = 4 A, M = 107.87 g/mol. So t = (0.25 g 1 96485 C/mol) / (4 A 107.87 g/mol). Let's calculate numerator: 0.25 96485 = 24121.25. Denominator: 4 107.87 = 431.48. Then t (in seconds) = 24121.25 / 431.48 ≈ 55.9 seconds, which is 55.9 / 60 ≈ 0.93 minutes. Wait, but that seems very fast. Wait, maybe the problem is 0.25 grams, and 4 A is 4 amps, which is 4 coulombs per second. So 55 seconds is about a minute. Maybe that's correct. Alternatively, maybe I made a mistake in the molar mass. Wait, 107.87 is correct. So the calculation seems right.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\boxed{0.93} (Wait, no, wait, let's check again. Wait, 0.25 g Ag: moles = 0.25 / 107.87 ≈ 0.002317 mol. Q = 0.002317 mol * 96485 C/mol ≈ 223.6 C. t = 223.6 / 4 = 55.9 s ≈ 0.93 min. But maybe the question has a typo, but according to the numbers, that's the answer. Alternatively, maybe I messed up the Faraday constant? No, 96485 C/mol is correct. So the answer is approximately 0.93 minutes. But let's check with exact calculation:
0.25 g Ag / 107.8682 g/mol = 0.0023175 mol Ag.
Moles of e⁻ = 0.0023175 mol.
Charge Q = 0.0023175 mol * 96485 C/mol = 223.6 C.
Time t = Q / I = 223.6 C / 4.0 A = 55.9 s.
Convert to minutes: 55.9 s / 60 s/min = 0.9317 min ≈ 0.93 min. So the answer is approximately 0.93 minutes.