QUESTION IMAGE
Question
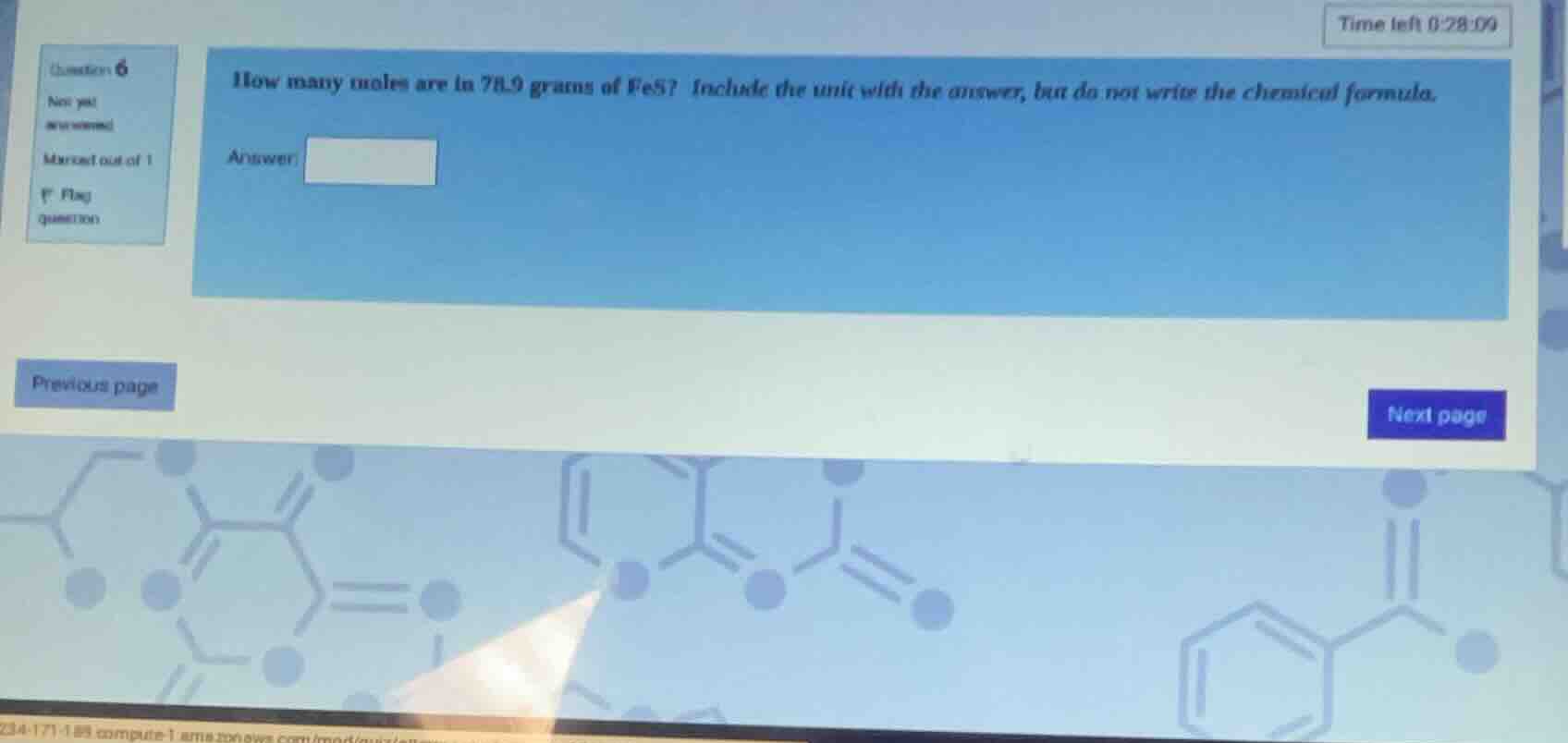
how many moles are in 78.9 grams of fes? include the unit with the answer, but do not write the chemical formula.
answer:
Step1: Calculate molar mass of FeS
Molar mass of Fe: $55.85\ g/mol$, molar mass of S: $32.07\ g/mol$.
Molar mass of FeS = $55.85 + 32.07 = 87.92\ g/mol$.
Step2: Use moles formula ($n = \frac{m}{M}$)
Given mass $m = 78.9\ g$, molar mass $M = 87.92\ g/mol$.
$n = \frac{78.9}{87.92} \approx 0.897\ mol$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$0.90\ mol$ (or $0.897\ mol$, depending on precision)