QUESTION IMAGE
Question
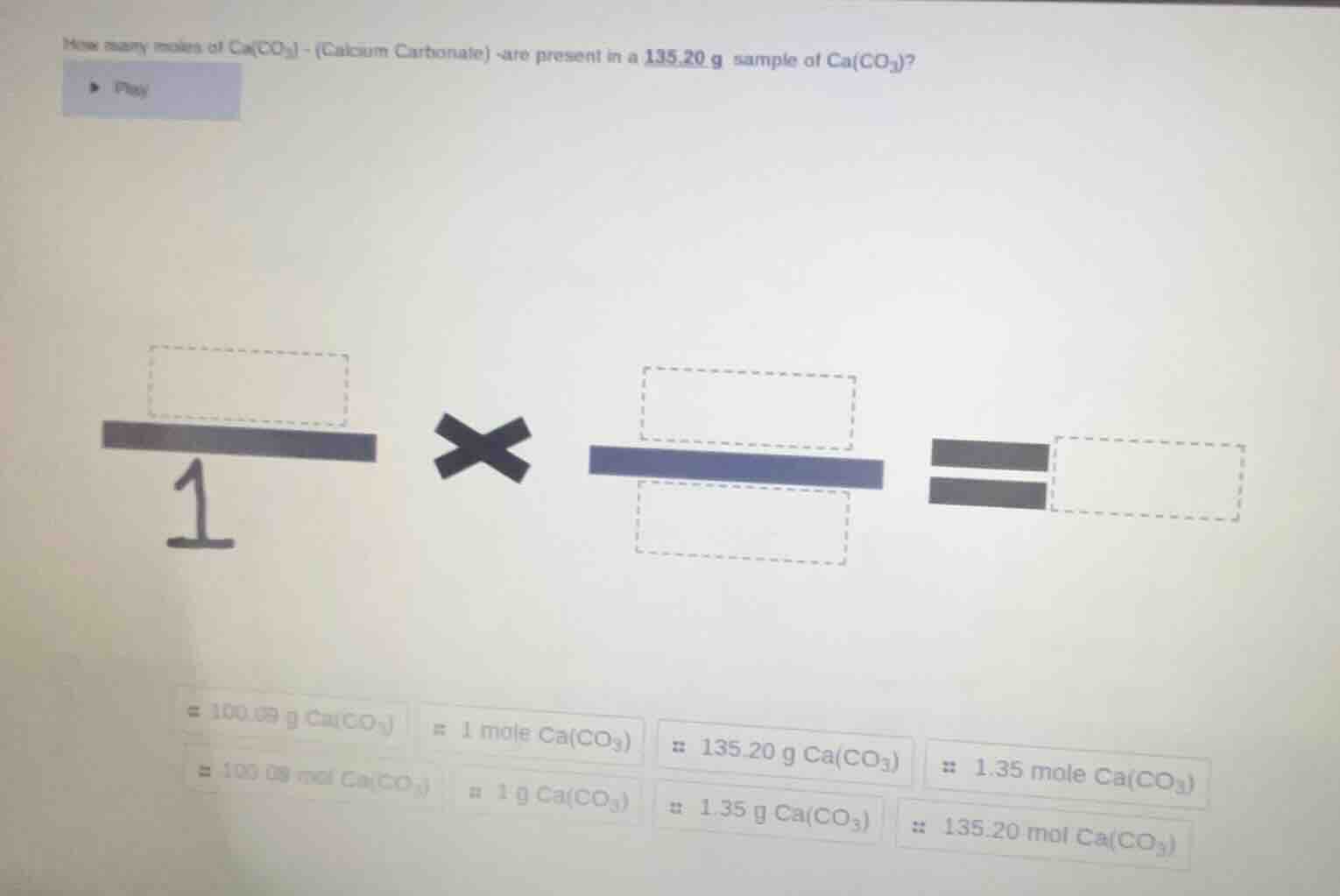
how many moles of ca(co₃) - (calcium carbonate) are present in a 135.20 g sample of ca(co₃)?
Step1: Identify given mass and molar mass
We know the mass of \( \text{Ca(CO}_3\text{)} \) is \( 135.20 \, \text{g} \), and the molar mass of \( \text{Ca(CO}_3\text{)} \) is \( 100.09 \, \text{g/mol} \) (calculated as \( 40.08 + 12.01 + 3\times16.00 = 100.09 \, \text{g/mol} \) for \( \text{Ca} \), \( \text{C} \), \( \text{O} \) respectively).
Step2: Use the formula \( n=\frac{m}{M} \) (moles = mass / molar mass)
The formula to calculate moles is \( n = \frac{m}{M} \), where \( m = 135.20 \, \text{g} \) and \( M = 100.09 \, \text{g/mol} \). Plugging in the values: \( n=\frac{135.20\,\text{g}}{100.09\,\text{g/mol}}\approx1.35\,\text{mol} \). In the given format, we multiply the mass (\( 135.20 \, \text{g} \)) by the conversion factor \( \frac{1\,\text{mol}}{100.09\,\text{g}} \), so \( 135.20\,\text{g} \times \frac{1\,\text{mol}}{100.09\,\text{g}} \approx 1.35\,\text{mol} \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The number of moles of \( \text{Ca(CO}_3\text{)} \) is \( 1.35 \) moles (or more precisely, using the given molar mass approximation \( \frac{135.20}{100.09}\approx1.35 \)). Filling in the boxes: First box (mass): \( 135.20 \, \text{g} \, \text{Ca(CO}_3\text{)} \), second box (numerator of conversion factor): \( 1 \, \text{mole} \, \text{Ca(CO}_3\text{)} \), third box (denominator of conversion factor): \( 100.09 \, \text{g} \, \text{Ca(CO}_3\text{)} \), and the result: \( 1.35 \, \text{mole} \, \text{Ca(CO}_3\text{)} \). So the calculation is \( 135.20\,\text{g} \times \frac{1\,\text{mol}}{100.09\,\text{g}} = 1.35\,\text{mol} \) (approx).