QUESTION IMAGE
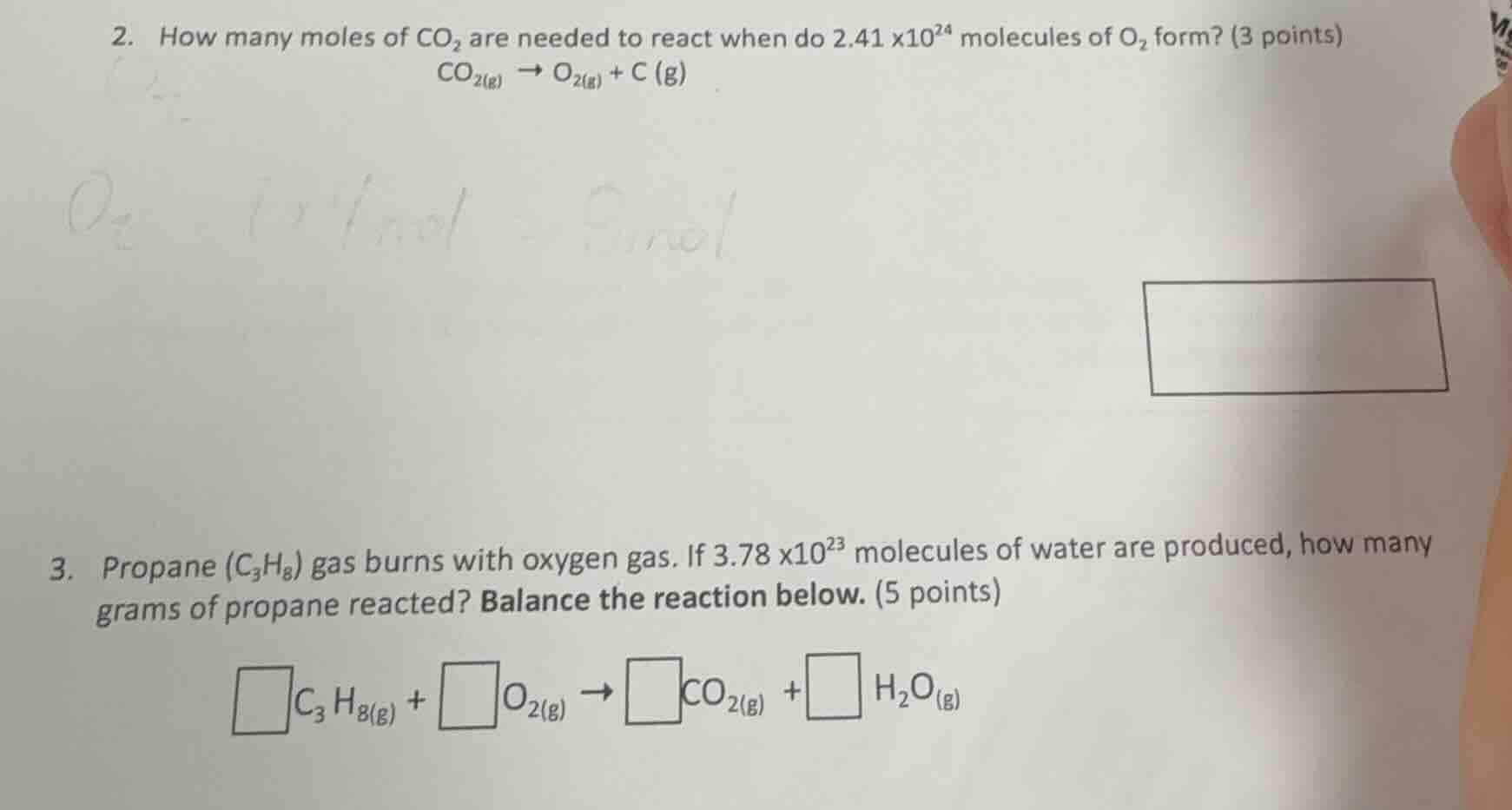
Question
- how many moles of co₂ are needed to react when do 2.41×10²⁴ molecules of o₂ form? (3 points)
co₂(g) → o₂(g) + c (g)
- propane (c₃h₈) gas burns with oxygen gas. if 3.78×10²³ molecules of water are produced, how many grams of propane reacted? balance the reaction below. (5 points)
□c₃h₈(g) + □o₂(g) → □co₂(g) + □ h₂o(g)
Question 2
Step1: Find moles of \( O_2 \)
Using Avogadro's number (\( 6.022 \times 10^{23} \) molecules/mol), moles of \( O_2 = \frac{2.41 \times 10^{24}\ \text{molecules}}{6.022 \times 10^{23}\ \text{molecules/mol}} \approx 4.00\ \text{mol} \).
Step2: Use stoichiometry (balanced equation \( \text{CO}_2
ightarrow \text{O}_2 + \text{C} \) has 1:1 ratio for \( \text{CO}_2 \) and \( \text{O}_2 \))
Moles of \( \text{CO}_2 \) = moles of \( O_2 = 4.00\ \text{mol} \).
Step1: Balance the equation
For \( \text{C}_3\text{H}_8 + \text{O}_2
ightarrow \text{CO}_2 + \text{H}_2\text{O} \):
- C: 3 on left, so \( 3\ \text{CO}_2 \).
- H: 8 on left, so \( 4\ \text{H}_2\text{O} \) (8 H).
- O: \( 3 \times 2 + 4 \times 1 = 10 \) O on right, so \( 5\ \text{O}_2 \) (10 O).
Balanced: \( 1\ \text{C}_3\text{H}_8 + 5\ \text{O}_2
ightarrow 3\ \text{CO}_2 + 4\ \text{H}_2\text{O} \).
Step2: Moles of \( \text{H}_2\text{O} \)
\( \text{Moles} = \frac{3.78 \times 10^{23}\ \text{molecules}}{6.022 \times 10^{23}\ \text{molecules/mol}} \approx 0.628\ \text{mol} \).
Step3: Moles of \( \text{C}_3\text{H}_8 \) (1:4 ratio from balanced equation)
\( \text{Moles of } \text{C}_3\text{H}_8 = \frac{0.628\ \text{mol}\ \text{H}_2\text{O}}{4} \approx 0.157\ \text{mol} \).
Step4: Mass of \( \text{C}_3\text{H}_8 \) (molar mass \( = 3(12.01) + 8(1.008) = 44.104\ \text{g/mol} \))
\( \text{Mass} = 0.157\ \text{mol} \times 44.104\ \text{g/mol} \approx 6.92\ \text{g} \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 4.00\ \text{moles} \)