QUESTION IMAGE
Question
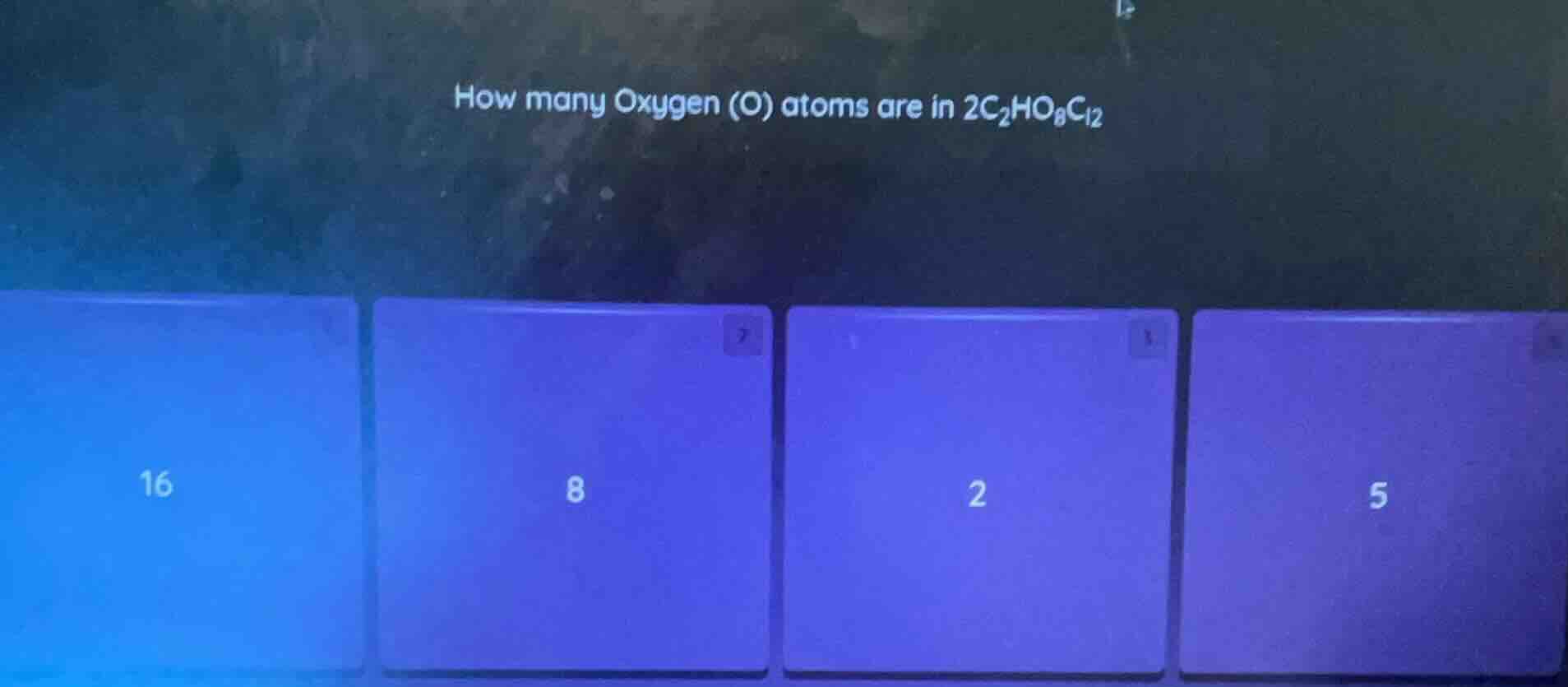
how many oxygen (o) atoms are in $2c_2ho_8cl_2$
options: 16, 8, 2, 5
Step1: Identify O atoms in one molecule
In the formula \( C_2HO_8Cl_2 \), the subscript of O is 8, so one molecule has 8 O atoms.
Step2: Multiply by the coefficient
The coefficient in front of the formula is 2, so total O atoms = \( 2 \times 8 = 16 \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
16